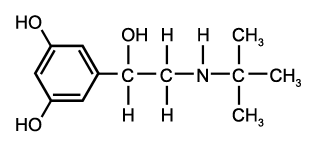
Study objective: Aerosolized [[Beta].sub.2]-agonists have been associated with increased morbidity in asthmatics. These drugs cause transient increases in heart rate and decreases in serum potassium levels after these drugs are first utilized. This study is designed to elucidate whether [Beta]-adrenergic tolerance to the hemodynamic, cardiac, and electrolyte effects of inhaled terbutaline occurs during 14 days of maintenance therapy.
Design: Eight patients with stable asthma weaned off [[Beta].sub.2]-agonist therapy were studied in a randomized, double-blinded, placebo-controlled study utilizing aerosolized terbutaline, 400 [micro] g q6h. Hemodynamic measurements and M-mode echocardiography were performed before and 15 and 30 min after the initial dose of terbutaline or placebo and after a dose of aerosolized terbutaline after 14 days of aerosolized terbutaline maintenance therapy. Holter monitors were worn on the first day of placebo or terbutaline therapy and on day 14 of terbutaline therapy. Plasma potassium, bicarbonate, and glucose levels, pH, renin activity, and serum insulin and aldosterone levels were measured before and after 24 and 48 h after terbutaline or placebo therapy and after 14 days of aerosolized terbutaline maintenance therapy.
Results: Terbutaline increased cardiac index and decreased systemic vascular resistance greater after 14 days of therapy compared with the first dose (5.2 [+ or -] 0.5 vs 4.4 [+ or -] 0.6 L/min/[m.sup.2]; p [is less than] 0.05; and 760 [+ or -] 62 vs 1,016 [+ or -] 118 dyne [multiplied by] s [multiplied by] [cm.sup.-5], p [is less than] 0.01). After 14 days of terbutaline therapy, the mean maximum heart rate and number of episodes of heart rate [is greater than] 100 beats/mm were higher compared with the other study day (p [is less than] 0.05). Plasma potassium level decreased from 4.29 [+ or -] 0.09 to 3.65 [+ or -] 0.16 mmol/L after 24 h of terbutaline and to 3.90 [+ or -] 0.11 mmol/L after 48 h. Plasma potassium level returned to baseline after 14 d of terbutaline therapy. Plasma glucose and serum insulin levels rose significantly 24 h and 48 b after terbutaline and returned to baseline after 14 d of terbutaline therapy. Serum aldosterone level decreased significantly as serum potassium level decreased in the first 48 h of terbutaline therapy but returned to baseline levels after 14 d of terbutaline.
Conclusions: Cardiovascular [[Beta].sub.2]-receptors in patients with stable asthma do not develop tolerance to the effects of low-dose aerosolized terbutaline after 14 days of maintenance therapy. In contrast, the homeostatic mechanisms regulating serum potassium develop tolerance to low-dose terbutaline maintenance therapy. Lack of cardiovascular tolerance to maintenance doses of aerosolized [[Beta].sub.2]-agonists may be important in increased morbidity if excessive amounts of these drugs are administered during asthma exacerbations.
(CHEST 1998; 114:380--387)
Key words: [Beta]-adrenergic; cardiac; potassium; terbutaline
Abbreviations: MVCFS=maximum velocity of circumferential fiber shortening
Although aerosolized [[Beta].sub.2]-agonists are frequently utilized as maintenance therapy for asthma, this practice is now being questioned since [[Beta].sub.2]-agonists may worsen asthma control, especially on drug therapy withdrawal, or may be associated with increased asthma mortality.[1-4] Standard dosages of inhaled [[Beta].sub.2]-agonists in asthmatics produce a slight increase in heart rate and systolic BP and rarely induce ventricular or supraventricular premature contractions.[5-8] However, Chapman and colleagues[9] performed M-mode echocardiography in normal subjects after aerosolized fenoterol and showed that cardiac output increased 26% 30 rain after an inhaled dose, accompanied by an 18% fall in systemic vascular resistance. In addition, an 18% increase in the mean velocity of circumferential fiber shortening suggested an inotropic effect of aerosolized fenoterol. Thus, standard hemodynamic measurements, including changes in heart rate and BP after inhaled [[Beta].sub.2]-agonists, may underestimate the hemodynamic effects of these agents. Whether these cardiac and hemodynamic effects will persist during maintenance therapy with standard, low doses of inhaled [[Beta].sub.2]-agonists is unknown.
In addition, terbutaline may induce hypokalemia within minutes after drug administration by stimulating cellular potassium uptake, especially when administered by the IV or subcutaneous route.[10,11] This decrease in serum potassium level is transient since serum potassium levels return to near normal levels after 2 h.[11] The mechanisms for terbutaline-induced cellular potassium uptake have included an increase in glucose and insulin levels induced by [Beta]-adrenergic-mediated glycogenolysis or direct [[Beta].sub.2]-receptor stimulation of insulin release from the pancreas.[10-14] The latter mechanism is supported by the finding that somatostatin infusion during terbutaline therapy inhibited pancreatic insulin release and prevented a decrease in serum potassium level, presumably by diminishing cellular potassium uptake.[15] In contrast, Tveskov and colleagues[16] suggested that IV terbutaline induces significant increases in the number of sodium/potassium pumps in muscle cells, suggesting that terbutaline mediates cellular potassium uptake independent of increases in glucose and insulin levels. Moreover, to our knowledge, no studies have analyzed the effects of inhaled [[Beta].sub.2]-agonists, especially terbutaline, on other parameters that affect potassium homeostasis such as the renin-angiotensin-aldosterone system or changes in acid-base homeostasis. Indeed, [Beta]-adrenergic stimulation has been shown to stimulate renin release, which leads to increases in plasma aldosterone levels, which can promote cellular potassium uptake or the renal excretion of potassium.[17,18] In addition, [Beta]-adrenergic-mediated glycogenolysis in muscle has been shown to produce lactic acidosis, which could attenuate cellular potassium uptake.[19]
The purpose of this study is to determine the hemodynamic and cardiac effects of inhaled terbutaline administered in standard aerosolized doses after exposure to the first dose and after 2 weeks of maintenance therapy. This study utilizes echocardiography to assess the hemodynamic and cardiac effects of inhaled terbutaline to determine whether cardiac [Beta]-receptor desensitization occurs after sustained exposure to low doses of terbutaline. In addition, we assessed the electrolytes' effects of standard doses of inhaled terbutaline after 24 and 48 h to determine whether more prolonged therapy with aerosolized terbutaline affects potassium homeostasis and to define the mechanisms of any hypokalemic effect demonstrated.
MATERIALS AND METHODS
Eight nonsmoking, asthmatic patients, 26 to 43 years old (mean, 32 years) were studied in a double-blind, randomized, crossover design. Patients were enrolled after giving informed consent to this study that was approved by the institutional review board. Asthma was defined in each patient by demonstrating reversible obstructive pulmonary, disease quantitated as [is greater than or equal to] 20% improvement in [FEV.sub.1] after bronchodilators. Each patient was screened to ensure that M-mode and two-dimensional echocardiograms of high technical quality could be obtained. All patients had been treated in the past with inhaled [[Beta].sub.2]-agonists prior to entering this study and were skilled in the use of aerosolized inhalers. No patient received any inhaled [[Beta].sub.2]-agonists for 2 weeks prior to this study. The patients abstained from caffeine-containing beverages for 12 h and from alcohol for 24 h before any pulmonary and echocardiographic testing. No caffeine or alcohol was ingested during 24-h Holter monitoring. Maintenance therapy with either theophylline or inhaled glucocorticoid drugs was discontinued 48 h prior to entering the study except for two patients who were maintained on regimens of stable daily doses of oral prednisone (5 mg and 10 mg) throughout the entire study period.
Baseline studies included spirometry to assess pulmonary function, 24-h Holter monitoring, and an ECG. Training sessions on the proper administration of metered-dose inhaler utilizing 'spacer devices were performed before initiation of the study. Patients were then randomized to receive either terbutaline sulfate (Brethaire; Ciba-Geigy Corp; Summit, NJ) at a dosage of 400 [micro] g q6h for 48 h or placebo for 48 h followed by the other agent for 48 h. Thereafter, each patient received terbutaline at a dosage of 400 [micro] g q6h for 14 days from a metered-dose inhaler containing 75 mg of terbutaline capable of delivery of 300 actuations. Patients were instructed to self-administer two puffs, 1 rain apart, of either terbutaline or placebo via a metered-dose inhaler utilizing a spacer device (Breathancer; Ciba-Geigy Corp). All measurements and blood samples were obtained 6 h after a standardized breakfast and in a darkened, quiet room with the patient in supine position and at rest. On the first day of either terbutaline or placebo and after 14 days of inhaled terbutaline therapy, a 24-h Holter monitoring recording was performed. M-mode and two-dimensional echocardiography were performed before and 1.5 and 30 min after the initial 400-[micro] g dose of terbutaline and placebo and 14 days later after a 400-[micro] g dose of terbutaline. Supine and standing BP and heart rate were obtained before and 15, 30, and 45 rain after terbutaline on the echocardiography days. The terbutaline dose inhalers utilized for 14 days were retrieved and tested for the number of actuations remaining to assure compliance with the protocol. Approximately one third of each inhaler was utilized, which closely approximates the correct 14 days of aerosolized terbutaline.
Echocardiograms were performed (MK-600 Cardioseries Ultrasound System; Advanced Technologies Lab, Inc; Bellevue, Wash). The machinery utilized (ATL 860-C, an ATL Adaptive Doppler Flow Analyzer, an(] an ATL Echoanalyzer, 51-D) allowed both M-mode and two-dimensional analysis. The two-dimensional echocardiograms allowed optimal placement for M-mode echocardiography. Holter monitoring utilized a specific monitor (Model 44-B Holter Monitor; Delmar Avionics Electrocardiocoder; Delmar, Calif). The tracings were analyzed using an analyzer (Delmar Avionics Automated Model CR-100 Cardioexpress Analyzer). All Holter tracings were analyzed blindly by one of the investigators. Systemic vascular resistance, percent fractional shortening, left ventricular ejection fraction, mean velocity of circumferential fiber shortening (MVCFS), cardiac output, and cardiac index were calculated using standard formulas. The maximum velocity of circumferential fiber shortening was normalized for changes in heart rate.[20] Hemodynamic measurements were obtained using a sphygmomanometer (Tycos; Arden, NC) that was calibrated each week.
On the first day of either terbutaline or placebo therapy and on day 14 of terbutaline therapy, pulmonary function tests utilizing a spirometer (Puritan-Bennett Spirometer, PS-600; Baltimore) were performed before and 45 rain after a 400-[micro] g dose of terbutaline or placebo on the placebo day. The best two of three measurements were averaged for the analysis.
To assess the effects of terbutaline and placebo on potassium homeostasis, blood samples were obtained before starting treatment with either agent, after 24 and 48 h of either terbutaline or placebo therapy, and on day 14 of terbutaline therapy. Samples were analyzed for plasma sodium, potassium, chloride, bicarbonate, glucose, lactate, renin activity, venous pit, serum insulin, and serum aldosterone. Plasma sodium and potassium levels were measured by flame photometry on a photometer (Instrumentation Lab Flame Photometer, Model 143; Instrumentation Labs; Lexington, Mass). Plasma glucose, chloride, and bicarbonate levels were measured on a spectrophotometer (Gilford 103 Automated Spectrophotometer; Oberlin, Ohio). Venous pit was measured on a blood gas analyzer (Instrumentation Laboratory pH Blood Gas Analyzer 813; Lexington, Mass). Aldosterone was measured on ethyl acetate extracts of plasma after chromatography on celite-ethylene glycol microcolumns. Radioimmunoassay was performed using aldosterone -1, 2, 6, 7-[H.sup.3] (New England Nuclear Corp; North Billerica, Mass) and a sensitive sheep antialdosterone antibody (Radioassay Systems Laboratories; Carson, Calif). Separation of unbound aldosterone was performed with a dextran-charcoal suspension. Sensitivity was 5 pg per tube or 1.5 ng/dL. Assay recoveries were 93 to 97% using [H.sup.3] aldosterone spiked serum. Interassay and intra-assay precision was 6% and 9.6% CV, respectively. The normal adult reference range is 2 to 20 ng/dL. Plasma renin activity was estimated using a radioimmunoassay kit (Rianen Angiotensin I Radioimmunoassay Kit; New England Nuclear Corp). Recovery of added angiotensin I was 95 to 100%. Intra-assay precision was 8.7% and 5.2% CV for low and high plasma pool replicators. Sensitivity was approximately 30 pg/mL. Plasma renin activity is expressed as ng of angiotensin I generated per mL/h. The normal adult reference range is 20 to 160 ng/mL/h. Insulin was measured (using the "Coat-a-count" Insulin Radioimmunoassay Kit; Diagnostic Products Corp; Los Angeles). The assay is performed on undiluted plasma in antibody-coated tubes. Sensitivity was approximately 2 [micro]U/mL and intra-assay precision was 7%. Recovery from insulin-spiked sera was 100%. The normal adult reference range was 0 (unmeasurable) to 30 uU/mL.
Statistical Analysis
Data were analyzed utilizing the Student's t test for paired data and by analysis of variance. A p value [is less than] 0.05 was considered significant. Since there were no differences between the two groups who initially received either placebo or terbutaline for 48 h, both groups were added together for statistical analysis.
RESULTS
Hemodynamics
Supine heart rate rose slightly 15 rain after the initial dose of inhaled terbutaline (Table 1) and returned to baseline after 30 min. No changes in systolic or diastolic BP or mean arterial BP were found. After 14 days of terbutaline therapy, the pulse rate increased slightly after two puffs of terbutaline. No changes in any hemodynamic parameters were noted after placebo. In addition, no changes in standing pulse rate or BP were noted after the initial dose of terbutaline or after 14 days of terbutaline therapy.
[TABULAR DATA 1 NOT REPRODUCIBLE IN ASCII]
Echocardiography
A decrease in systemic vascular resistance 30 rain after the initial two puffs of terbutaline was associated with a significant increase in cardiac index (Table 1). The decrease in systemic vascular resistance and increase in cardiac index were accentuated after 14 days of inhaled terbutaline therapy. Systemic vascular resistance progressively decreased 15 and 30 rain after terbutaline therapy, which correlated with a progressive increase in cardiac index (Table 1). After 14 days of terbutaline therapy, the mean cardiac index 30 rain after terbutaline was significantly greater at 5.2 [+ or -] 0.5 L/min/[m.sup.2] compared with just 4.4 [+ or -] 0.6 L/min/[m.sup.2] after the first inhaled terbutaline dose (p [is less than] 0.05). After 14 days of terbutaline therapy, the mean systemic vascular resistance 30 min after terbutaline was significantly lower compared with 30 min after the first close of terbutaline (760 [+ or -] 69, vs 1,016 [+ or -] 118 dyne [multiplied by] s [multiplied by] [cm.sup.-5]; p [is less than] 0.01). There were no changes in any echocardiographic measurements with placebo. In addition, 15 rain after the initial two puffs of terbutaline, the absolute and normalized MVCFS decreased but returned to baseline after 30 rain (Table 1). In contrast, after 14 days of maintenance terbutaline therapy, the MVCFS increased significantly both 15 and 30 rain after inhaled terbutaline. However, when the MVCFS was normalized for increases in heart rate after terbutaline,[20] there was no significant change in MVCFS, indicating no true increase in ventricular contractility (Table 1).
Holter Monitoring
One patient developed ventricular premature beats during the last day of terbutaline therapy, and one patient developed atrial premature beats during placebo therapy and on the first day of terbutaline therapy but not on the 14th day of terbutaline therapy. No one developed supraventricular or ventricular tachycardia. After 14 days of terbutaline therapy, the mean number of episodes of heart rate [is greater than] 100 beats/min was higher when compared with the other study days, p [is less than] 0.05 (Table 2). In addition, the mean maximal pulse rate was greater on day 14 of terbutaline therapy when compared with the other study days, p [is less than] 0.05 (Table 9,).
(*) Data expressed as mean [+ or -] SE.
([dagger]) p<0.05 compared with all other days.
Electrolyte, Acid Base, and Hormonal Changes
A significant decrease in plasma potassium level was noted both 24 and 48 h after terbutaline therapy (Fig 1). The mean plasma potassium level decreased from 4.29 [+ or -] 0.09 mmol/L to 3.65 [+ or -] 0.16 mmol/L 24 h after terbutaline therapy (p [is less than] 0.005). At this time, four patients (one patient receiving prednisone) had a plasma potassium level [is less than] 3.70 mmol/L (range, 3.0 to 3.6 mmol/L). After 48 h of terbutaline therapy, all patients had a normal level of plasma potassium [is greater than] 3.70 mmol/L, but the mean level of 3.90 [+ or -] 0.11 mmol/L was still below the baseline level (p [is less than] 0.05). Despite the decreases in plasma potassium levels, no changes in cardiac rhythm were detected during Holter monitoring.
[Figure 1 ILLUSTRATION OMITTED]
Plasma glucose level rose from 83 [+ or -] 5 to 99 [+ or -] 6 mg/dL after 9.4 h of terbutaline therapy (p [is less than] 0.025). Serum insulin level increased after terbutaline therapy concomitantly with an increase in plasma glucose level (Fig 2). After 48 h of terbutaline therapy, the serum insulin level peaked at a level of 47 [+ or -] 13 uU/mL (p [is less than] 0.05 compared with baseline). Plasma glucose and serum insulin levels decreased after 14 days of terbutaline therapy and were not statistically different from baseline values. Data analysis, excluding the two patients receiving low-dose maintenance prednisone therapy, did not alter these results.
[Figure 2 ILLUSTRATION OMITTED]
No changes in plasma bicarbonate level or venous pH were noted throughout the study. A small increase in plasma lactate level was noted both 24 and 48 h after terbutaline therapy but not after 14 days of therapy (p [is less than] 0.05). Although these changes were statistically significant, the plasma lactate levels were still within the normal range of this assay method (0.5 to 2.9 mmol/L). No changes in plasma renin activity were found, but serum aldosterone level decreased significantly both 24 and 48 h after terbutaline therapy. The decrease in serum aldosterone level was associated with a concomitant decrease in plasma potassium level. Serum aldosterone levels returned to baseline after 14 days of terbutaline therapy. No changes in any of these parameters occurred during treatment with placebo.
Pulmonary Function
The [FEV.sub.1] increased significantly after the first inhalation of terbutaline (3.00 [+ or -] 0.21 to 3.26 [+ or -] 0.19, p [is less than] 0.001) and after 14 days of terbutaline therapy (3.09 [+ or -] 0.18 to 3.24 [+ or -] 0.20, p [is less than] 0.05) but not after placebo. Similar results were found for the forced expiratory flow rate between 25% and 75% of the FVC and peak expiratory flow.
DISCUSSION
Our study carefully examined the hemodynamic and cardiac effects of inhaled terbutaline after the initial dose and 2 weeks later after maintenance therapy by utilizing M-mode and two-dimensional echocardiography. The initial dose of terbutaline produced a transient increase in heart rate and cardiac output with a simultaneous decrease in systemic vascular resistance. After 2 weeks of maintenance terbutaline therapy, a significant increase in heart rate occurred and the decrease in systemic vascular resistance and increase in cardiac output after inhaled terbutaline were much greater than that demonstrated after the initial dose given 9, weeks earlier. These important physiologic changes could not be demonstrated by standard hemodynamic measurements, such as supine and standing heart and BP. Moreover, the number of episodes of heart rate [is greater than] 100 beats/rain and mean maximal heart rate were greater after 14 days of terbutaline therapy compared with the first day of terbutaline or placebo therapy. These findings suggest that low-dose maintenance therapy with aerosolized terbutaline in asthma patients does not produce high enough blood levels of terbutaline to down-regulate cardiac [Beta]-receptors. Chapman and colleagues[9] demonstrated similar findings utilizing M-mode echocardiography after therapeutic doses of inhaled fenoterol given to healthy individuals.
The cardiac effects of IV terbutaline in normal subjects, both with and without [[Beta].sub.1]-blockade by atenolol, have been studied.[21] Terbutaline increased heart rate and cardiac output and lowered systemic resistance and diastolic pressure similarly with or without atenolol pretreatment in normal individuals. However, these hemodynamic effects were enhanced by atropine pretreatment, suggesting that increases in vagal tone from terbutaline therapy attenuate the hemodynamic and cardiac effects of this agent.[22] Taken together, these studies suggest that the chronotropic effect of terbutaline is probably secondary to [[Beta].sub.2]-mediated increases in heart rate either due to direct stimulation of cardiac [[Beta].sub.2]-receptors or secondary to [[Beta].sub.2]-mediated vasodilation that induces reflex baroreceptor increases in heart rate. In contrast, larger quantities of terbutaline administered orally in healthy asthmatic subjects or in patients with COPD lead to higher blood and tissue terbutaline levels and cardiac [Beta]-receptor down-regulation.:[11,23-25] Our study demonstrates that patients with asthma initially respond to terbutaline with increases in cardiac output and decreases in systemic vascular resistance. These effects were significantly greater after 14 days of drug therapy. This suggests that peripheral vascular and/or cardiac [[Beta].sub.2]-adrenergic receptors in asthmatic patients do not down-regulate after repetitive stimulation by low-dose aerosolized terbutaline.
No physiologically important changes in myocardial contractility occurred. The increase in MVCFS after 14 days of therapy was due to increases in heart rate. After normalization of the MVCFS for increases in heart rate produced by terbutaline, no positive inotropic effect was found after initial exposure to or 14 days of terbutaline therapy. No significant arrhythmias occurred despite significant decreases in plasma potassium levels during the first 24 h of therapy. Numerous studies have demonstrated the lack of adverse effects of inhaled [Beta]-agonists on cardiac rhythm in asthmatic patients.[8]
[[Beta].sub.2]-Adrenergic stimulation has the potential to affect potassium homeostasis by numerous mechanisms. First, terbutaline therapy may increase pancreatic insulin release either due to direct [[Beta].sub.2]-receptor stimulation of pancreatic islet cells[12] or due to increases in blood glucose levels resulting from [Beta]-adrenergic-mediated glycogenolysis and gluconeogenesis in the liver.[13,14] Increases in plasma insulin levels could stimulate cellular potassium uptake.[26] Moreover, [Beta]-adrenergic stimulation in muscle cells may directly induce cellular potassium uptake by producing an increase in intracellular cyclic adenosine monophosphate that stimulates cellular Na-K adenosine triphosphatase pumps.[16,26] In addition, plasma renin activity may increase from [Beta]-adrenergic stimulation with concomitant increases in plasma aldosterone levels that could either enhance renal potassium excretion in the distal nephron or exert a permissive effect on cellular potassium uptake.[17,18] Finally, [Beta]-adrenergic stimulation may induce lactic acidosis due to overproduction of lactic acid from [Beta]-adrenergic-induced muscle glycogenolysis, which could attenuate cellular potassium uptake.[19]
Recently, a subcutaneous injection of 250 [micro] g of terbutaline in asthmatic patients induced a significant decrease in serum potassium level as early as 15 min after administration that was associated with increases in both serum glucose and insulin levels.[10] The decrease in serum potassium returned to baseline levels after 2 h along with a concomitant return of glucose and insulin levels to baseline levels. Additional studies utilizing somatostatin to inhibit pancreatic insulin release attenuated the decrease in serum potassium level from IV terbutaline.[15] Taken together, these studies suggest that insulin-mediated cellular potassium uptake is the proximate cause for hypokalemia induced by terbutaline. In contrast, pharmacologic doses of terbutaline may increase the number of NA-K adenosine triphosphatase pumps in muscle cells that could stimulate cellular potassium uptake.[16]
Our study evaluated the effects of aerosolized terbutaline on potassium homeostasis after more prolonged administration. After 24 and 48 h of low-dose inhaled terbutaline therapy, plasma potassium levels significantly decreased compared with baseline. There were significant increases in plasma glucose and serum insulin levels at both times. In contrast, there were no significant changes in plasma potassium, plasma glucose, or serum insulin levels after 14 days of aerosolized terbutaline therapy. Serum insulin levels 24 and 48 h after terbutaline therapy were at a level (40 [micro] U/mL) that can enhance cellular potassium uptake.[26] Thus, our study extends earlier observations by demonstrating that increases in blood glucose and insulin levels play a role in terbutaline-induced hypokalemia during the first 48 h of maintenance therapy. However, plasma potassium returned to the baseline level after 14 days, possibly due to down-regulation of muscle and liver [Beta]-receptors responsible for cellular potassium uptake after more prolonged therapy. It is possible that tachyphylaxis to the hypokalemic effects of terbutaline could occur after 14 days of therapy. Indeed, Lipworth and colleagues[27] showed that increasing repetitive doses of aerosolized terbutaline on the same day in patients with chronic lung disease produced a decrease in plasma potassium level of only 0.09 mmol/L after 14 days of low-dose inhaled terbutaline therapy.[27] However, bronchodilator tachyphylaxis after low-dose or high-dose maintenance terbutaline therapy did not occur in this study or in our study utilizing low-dose terbutaline therapy.[27] In contrast, these same investigators did not find tachyphylaxis to the hypokalemic effects of low-dose aerosolized albuterol but did demonstrate tachyphylaxis to high-dose maintenance therapy with albuterol.[28] In addition, earlier studies found no significant increases in urinary potassium excretion to explain the decrease in blood potassium level induced by terbutaline.[10,15]
Our study also examines the effects of inhaled [Beta]-agonists on lactate production and changes in plasma renin activity and serum aldosterone levels. Although we found a statistically significant increase in plasma lactate levels after both 24 and 48 h of aerosolized terbutaline therapy, these levels were within the normal range. No significant decrease in plasma bicarbonate level or venous pH was found that could attenuate cellular potassium uptake. In addition, there was no significant change in plasma renin activity, but there was a significant decrease in serum aldosterone level concomitant with the decrease in plasma potassium level. Thus, increases in serum aldosterone levels, which can have a permissive effect on cellular potassium uptake[17,18] or renal potassium excretion, did not contribute to terbutaline-induced hypokalemia.
In summary, our study demonstrates that aerosolized terbutaline can have important hemodynamic effects that are missed by standard measurements of BP and heart rate. Significant increases in cardiac output with concomitant decreases in systemic resistance occur after initial exposure to aerosolized terbutaline, and these changes are enhanced after 14 days of therapy. In contrast, we have shown that changes in blood potassium, glucose, and insulin levels that occur after aerosolized terbutaline therapy persist for up to 24 to 48 h but-return to normal after 14 days of therapy. These observations suggest that cardiac and/or vascular [Beta]-receptors may be less susceptible to down-regulation after repetitive stimulation than [Beta]-receptors in skeletal muscle or liver. Lack of cardiovascular tolerance to maintenance doses of aerosolized [[Beta].sub.2]-agonists may be important in increased morbidity if excessive amounts of these drugs are administered during asthma exacerbation.[5] However, we demonstrated that aerosolized terbutaline administered in standard maintenance doses appears to be safe in patients with stable asthma.
ACKNOWLEDGMENTS: We would like to thank Ann Vasseur, Walter Stupack, and David Watson for their assistance in the laboratory measurements; Joyce Dugre for assistance in performing the echocardiograms; and Meri Kassanos for assistance in typing this manuscript.
REFERENCES
[1] Barrett TE, Strom BL. Inhaled beta-adrenergic receptor agonists in asthma: more harm than good? Am J Respir Care Med 1995; 151:574-77
[2] McFadden ER Jr. Perspectives in [[Beta].sub.2]-agonist therapy: vox clamantis in deserto vel lux in tenebris? J Allergy Clin Immunol 1994; 95:641-51
[3] Devoy MAB, Fuller RW, Palmer JBD. Are there any detrimental effects of the use of inhaled long-acting 132 agonists in the treatment of asthma? Chest 1995; 107:1116-24
[4] Jenne JW, Tashkin DP. Beta-adrenergic agonists: physiology and pharmacology. In: Weiss EB, Stein M, eds. Bronchial asthma: mechanisms and therapeutics. Boston: Little Brown, 1993; 700-48
[5] Wahedna I, Wong CS, Wisniewski AFZ, et al. Asthma control during and after cessation of regular [[Beta].sub.2]-agonist treatment. Am Rev Respir Dis 1993; 148:707-12
[6] Carlstrom S, Westling H. Metabolic, circulatory and respiratory effects of a new sympathomimetic [Beta]-receptor-stimulating agent, terbutaline, compared with those of orciprenaline. Acta Med Scand 1970; S512:33-40
[7] Wong CS, Pavord ID, Williams J, et al. Bronchodilator, cardiovascular, and hypokalaemic effects of fenoterol, salbutamol, and terbutaline in asthma. Lancet 1990; 336:1396-99
[8] Sessler CN, Ayres SM, Glauser FL. Cardiac interactions, arrhythmias, and pathology. In: Weiss EB, Stein M, eds. Bronchial asthma: mechanisms and therapeutics. Boston: Little Brown, 1993; 1045-53
[9] Chapman KB, Smith DL, Rebuck AS, et al. Hemodynamic effects of an inhaled beta-2 agonist. Clin Pharmacol Ther 1984; 35:762-67
[10] Kung M, White JR, Burki NK. The effect of subcutaneously administered terbutaline on serum potassium in asymptomatic adult asthmatics. Am Rev Respir Dis 1984; 129:329-32
[11] Bengtsson B, Fagerstrom PO. Extrapulmonary effects of terbutaline during prolonged administration. Clin Pharmacol Ther 1982; 31:726-32
[12] Cano A, Tovar I, Parrilla JJ, et al. Metabolic disturbances during intravenous use of ritodrine: increased insulin levels and hypokalemia. Obstet Gynecol 1984; 65:356-60
[13] Hems DA, Whitton PD. Control of hepatic glycogenolysis. Physiol Rev 1980; 60:1-50
[14] Sherwin RS, Sacca L. Effect of epinephrine on glucose metabolism in humans: contribution of the liver. Am J Physiol 1984; 247:E157-65
[15] Schnack C, Podolsky A, Watzke H, et al. Effects of somatostatin and oral potassium administration on terbutaline-induced hypokalemia. Am Bev Respir Dis 1989; 139:176-80
[16] Tveskov C, Djurhuus MS, Klitgaard H, et al. Potassium and magnesium distribution, ECG changes and ventricular ectopic beats during [~2-adrenergic stimulation with terbutaline in healthy subjects. Chest 1994; 106:1654-59
[17] Manolagas SC, Deftos LJ. Benin release [[Beta].sub.1] - or [[Beta].sub.2]-receptor mediated? N Engl J Med 1985; 312:19.3-24
[18] Bia MJ, Defronzo R. Regulation of extrarenal potassium homeostasis by adrenal hormones in rats. Am J Physiol 1982; 242:F641-49
[19] Kuo SH, Kamaka JK, Lum BKB. Adrenergic receptor mechanisms involved in the hyperglycemia and hypelactic-acidemia produced by sympathomimetic amines in the cat. J Pharmacol Exp Ther 1977; 202:301-09
[20] Knapp WH. Relationships between mean velocity of circumferential fiber shortening and heart rate-the diagnostic value of a normalization of VCF to heart rate. JCU 1978; 6:10-15
[21] Strauss MH, Reeves RA, Smith DL, et al. The role of cardiac beta-1 receptors in the hemodynamic response to a beta-2 agonist. Clin Pharmacol Ther 1986; 40:108-15
[22] Levine MAH, Leenen FHH. Role of [[Beta].sub.1]-receptors and vagal tone in cardiac inotropic and chronotropic responses to a [[Beta].sub.2]-agonist in humans. Circulation 1989; 79:107-15
[23] Jenne JW, Chick TW, Strickland RD, et al. Subsensitivity of beta responses during therapy with a long-acting beta-2 preparation. J Allergy Clin Immunol 1977; 59:383-90
[24] Pierson DJ, Hudson LD, Stark K, et al. Cardiopulmonary effects of terbutaline and a bronchodilator combination in chronic obstructive pulmonary disease. Chest 1980; 77:176-82
[25] Stein M, Deegan R, Wood AJJ. Long-term exposure to [b.sub.2]-receptor agonist specifically desensitizes [Beta]-receptor-mediated venodilation. Clin Pharmacol Ther 1993; 54:187-93
[26] Zierler KL, Rabinowitz D. Effect of very small concentrations of insulin on forearm metabolism: persistence of its action on potassium and free fatty acids without its effect on glucose. J Clin Invest 1967; 43:950-62
[27] Lipworth BJ, Clark RA, Dhillon DP, et al. Comparison of the effects of prolonged treatment with low and high doses of inhaled terbutaline on beta-adrenoceptor responsiveness in patients with chronic obstructive pulmonary disease. Am Rev Respir Dis 1990; 142:338-42
[28] Lipworth BJ, Struthers AD, McDevitt DG. Tachyphylaxis to systemic but not to airway responses during prolonged therapy with high dose inhaled salbutamol in asthmatics. Am Rev Respir Dis 1989; 140:586-92
(*) From the Department of Medicine, Baystate Medical Center, Springfield, Mass; Tufts University School of Medicine, Boston; and the Pulmonary/Critical Care Division, Department of Medicine, University of Michigan, Ann Arbor, Mich.
This work was supported by a grant from the Ciba-Geigy Corporation.
Manuscript received January 23, 1997; revision accepted February 5, 1998.
Correspondence to: Gregory L. Braden, MD, Chief, Renal Division, Baystate Medical Center, 759 Chestnut St, Springfield, MA 01199
COPYRIGHT 1998 American College of Chest Physicians
COPYRIGHT 2000 Gale Group