METHOD OF PREPARATION
Note: This preparation should be done in a laminar airflow hood in a cleanroom or via isolation barrier technology by a validated aseptic compounding pharmacist using strict aseptic technique.
1. Calculate the required quantity of each ingredient for the total amount to be prepared.
2. Accurately weigh and/or measure each ingredient.
3. Add the polysorbate 80, alcohol and edetate disodium to about 75 mL of bacteriostatic water for injection and mix well.
4. Add the diclofenac sodium and stir until dissolved.
5. Add sufficient bacteriostatic water for injection to volume and mix well.
6. Sterile filter through an appropriate 0.2-µm filter into sterile vials.
7. Package and label.
PACKAGING
Package in tight, light-resistant containers.1
LABELING
Keep out of reach of children. Use only as directed.
STABILITY
If prepared from nonsterile powders and no sterility testing is done, a beyond-use date of 24 hours at room temperature, 3 days at refrigerated temperature or 45 days at
USE
This injection has been used in the treatment of mild-to-moderate inflammatory pain.
QUALITY CONTROL
Quality-control assessment can include weight/volume, physical observation, pH, specific gravity, osniolality, assay, color, clarity, paniculate matter, sterility and pyrogenicity.2,3
DISCUSSION
Diclofenac sodium injection is commercially available in other countries, but not in the United States. It is used in the treatment of inflammatory diseases (rheumatoid arthritis, osteoarthritis, ankylosing spondylitis, juvenile arthritis), dysmenorrhea and for its antipyretic effect. It is used topically in the treatment of actinic keratoses as a 3% gel.
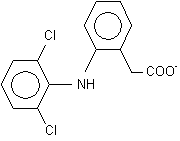
Diclofenac sodium (C^sub 14^H^sub 10^C^sub 12^NNaO^sub 2^, MW 318.13) is a phenylacetic acid derivative that is a nonsteroidal anti-inflammatory agent with antipyretic activity. It occurs as a white to off-white, hygroscopic, crystalline powder. It is soluble in ethanol and sparingly soluble in water. It melts at about 284°C. It should be preserved in tight, light-resistant containers.1,4
Polysorbate 80 (C^sub 64^H^sub 124^O^sub 26^, MW 1310, Tween 80, polyoxyethylene 20 sorbitan monooleate) occurs as a yellow oily liquid with a characteristic odor and a warm, somewhat bitter taste. It has a specific gravity of 1.06-1.09 and a hydrophile:lipophile balance (HLB) value of 15.0.5
Alcohol (C^sub 2^H^sub 5^OH, MW 46.07, ethyl alcohol, ethanol, grain alcohol) is a clear, colorless mobile and volatile liquid with a slight, characteristic odor and a burning taste. Alcohol USP refers to 95% ethanol. Its specific gravity is between 0.812 and 0.816, and its boiling point is 78.15°C. It is miscible with chloroform, glycerin and water, and its solutions may be sterilized by autoclaving or by filtration.6
Edetate disodium dihydrate (C^sub 10^H^sub 14^N^sub 2^Na2O^sub 8^, MW 336.21, edetate disodium, edetic acid disodium, disodium ethylenediaminetetraacetate) occurs as an odorless white crystalline powder with a slightly acid taste. It is soluble 1 g in 11 mL of water and is slightly soluble in 95% ethanol.6,8
Bacteriostatic water for injection (parabens) is prepared from water for injection that is sterilized and suitably packaged, containing one or more suitable antimicrobial agents. Bacteriostatic water for injection should be used with due regard for the compatibility of the antimicrobial agent or agents it contains with the particular medicinal substance that is to be dissolved or diluted. Bacteriostatic water for injection is a clear, colorless liquid, odorless or having the odor of the antimicrobial substance.1
REFERENCES
1. US Pharmacopeial Convention, Inc. United States Pharmacopeia 27-National Formulary22. Rockville, MD: US Pharmacopeial Convention, Inc.; 2004: 595-596, 1949, 2345-2349, 2759.
2. Allen LV Jr. Standard operating procedure for paniculate testing for sterile products. IJPC 1998; 2: 78.
3. Allen LV Jr. Standard operating procedure: Quality assessment for injectable solutions. IJPC 1999; 3: 406-407.
4. McEvoy GK. AHFS Drug Information-2004. Bethesda, MD: American Society of Health-System Pharmacists; 2004: 1958-1965, 3434-3435.
5. Lawrence MJ. Polyoxyethylene sorbitan fatty acid esters. In: Rose RC, Sheskey PJ, Weller PJ, eds. Handbook of Pharmaceutical Excipients. 4th ed. Washington, DC: American Pharmaceutical Association; 2003: 479-483.
6. Owen SC. Alcohol. In: Rowe RC, Sheskey PJ, Weller PJ, eds. Handbook of Pharmaceutical Excipients. 4th ed. Washington, DC: American Pharmaceutical Association; 2003: 13-15.
7. Owen SC. Edetic Acid. In: Rose RC, Sheskey PJ, Weller PJ, eds. Handbook of Pharmaceutical Excipients. 4th ed. Washington, DC: American Pharmaceutical Association; 2003: 225-228.
8. Reynolds JEF, ed. MARTINDALE: The Extra Pharmacopeia. 30 ed. London: Pharmaceutical Press; 1993: 681-682.
Copyright International Journal of Pharmaceutical Compounding Nov/Dec 2004
Provided by ProQuest Information and Learning Company. All rights Reserved