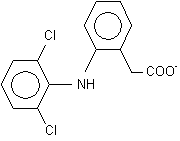
Abstract
A clearance rate of all occurrences [greater than or equal to]75% for actinic keratoses (AK) lesions is an accepted efficacy endpoint for topical agents. This efficacy endpoint has not been assessed for 3.0% diclofenac sodium gel (Solaraze[TM]).
We evaluated the efficacy and tolerability of 3.0% diclofenac sodium gel in the treatment of AK for a treatment period of 90 days and a 30-day follow-up period.
This is a multicenter, single-arm, open-label study in patients diagnosed with five or more AK lesions contained in 1 to 3 blocks (5 [cm.sup.2]) on the forehead, central face, or scalp. Patients were treated twice daily with a topical application of 3.0% diclofenac sodium gel for a period of 90 days with a follow-up assessment at 30 days post-treatment. The presence or absence of target lesions and new lesions was assessed at each visit a long with a global improvement index score.
Of the 76 patients who entered the study, 67 (88%) patients completed the study. At Day 90 of treatment, 78% of patients had [greater than or equal to]75% AK lesion clearance based on the target lesion number score (TLNS). Improving to 85% of patients demonstrating [greater than or equal to]75% AK lesion clearance at Day 120 (follow-up). Improvement was also demonstrated by 100% AK lesion clearance based on the TLNS clearance (Day 90 of treatment: 41%; Day 120 [follow-up]: 58%). Similar improvements were shown in cumulative lesion number score (CLNS), which included new as well as targeted AK lesions within the designated treatment areas, at Day 90 and Day 120 (follow-up). Investigators' assessment based on Investigator Global Improvement Index (IGII) confirmed the efficacy of 3.0% diclofenac gel in the clearance of AK lesions. A total of 39 patients (51%) experienced at least 1 adverse event considered to be related to 3.0% diclofenac sodium gel during the study. Dry skin and rash at the application site were most common reported adverse events, and most of these adverse events were mild or moderate in severity.
The topical application of 3.0% diclofenac sodium gel provides a safe and effective approach for the treatment of AK.
**********
Introduction
Actinic keratosis, or solar keratosis, is a pre-cancerous skin condition caused by excessive, prolonged exposure to ultraviolet light (1). The clinical features of AK lesions include rough, scaly patches, "bumps" on the skin, mottled skin, and cutaneous horns (2). These alterations in cell growth and differentiation can set the stage for evolution of AK lesions into invasive squamous cell carcinoma (SCC). In epidemiologic and histopathologic studies, AK lesions are the precursors of at least 60% of squamous cell cancers (3,4). As it is currently impossible to predict which lesions will progress to invasive SCCs, early and effective treatment is essential in reducing the risk of developing skin cancer in the longer term (5). SCC may account for up to 34% of deaths from skin cancer among adults to 84 years of age, and 56% of deaths among persons 85 years and older (6). Additionally, the morbidity and cost associated with SCC therapy is essential (7).
Destruction of AK lesions is the most commonly performed outpatient dermatological procedure in the United States. A National Ambulatory Medical Care Survey from 1993 and 1994 estimated that there were 3.7 million office visits and 5.2 million procedures for AK (more than 3 times the total office visits for squamous cell cancer, basal cell cancer, and melanoma combined) (8). The actual occurrence of AK lesions is likely to be much higher, as only a small proportion of individuals with AK lesions seek or receive treatment (9). The lesions of AK are most likely to appear in individuals over the age of 40, but in geographic areas with year-round high-intensity sunlight AK lesions are now found in persons as young as the teens and 20's. Due to the link between exposure to sunlight and the later development of actinic keratosis, people who have spent significant periods of their life outdoors appear to be at highest risk.
Treatment for AK lesions has traditionally involved painful procedures including cryosurgery, surgical excision and biopsy (for lesions suspected of being cancerous), and topical chemotherapy. As a first-in-class therapy, 3.0% diclofenac sodium gel provides an alternative topical treatment for AK lesions.
Randomized, double-blind, vehicle-controlled clinical trials have demonstrated the effectiveness of 3.0% diclofenac sodium gel in the clearance of 100% of AK lesions when applied for 60-90 days (10,11). These trials also demonstrated the tolerability of 3.0% diclofenac sodium gel, which has a low systemic absorption profile. This five-center, single-arm, open-label study was conducted to assess the efficacy and tolerability of 3.0% diclofenac sodium gel in the treatment of AK.
Methods and Patients
Study Design and Clinical Assessments
This was a multicenter, single-arm, open-label study. Patients were eligible for enrollment into the study at five centers in the USA if they met the inclusion and exclusion criteria, and the patient and physician agreed that treatment with 3.0% diclofenac sodium gel was an appropriate form of therapy. The treatment period was 90 days with monthly clinic visits (Days 30, 60, and 90), and a follow-up visit 30 days post-treatment (Day 120). Written informed consent was obtained from each patient prior to screening procedures.
Demographic and baseline characteristics including severity of disease (Baseline Severity Index [BSI]) and skin type were obtained at the screening/enrollment visit. The BSI is an investigator assessment of lesion severity on a scale of 1 to 3: 1 (mild) = Clearly visible lesions with mostly thin scales, which may be palpated; 2 (moderate) = Many visible, small lesions easily felt on palpation; and 3 (severe) = Many thick, hypertrophic lesions, which were clearly visible and palpable with well-defined borders. Skin type was evaluated on a scale of I (always burns easily; never tans) to IV (never burns; deeply pigmented) based on the patient's sun burning and tanning history. The efficacy of the treatment in providing clearance of AK lesions was assessed using two quantitative measures and one qualitative measure. The quantitative measurements comprised the Target Lesion Number Score (TLNS) and the Cumulative Lesion Number Score (CLNS), and were determined at each post-baseline clinic visit. The TLNS consisted of the total number of lesions identified in up to three designated 5 [cm.sup.2] treatment blocks at baseline. Treatment blocks were monitored at all subsequent clinic visits to measure the number of lesions cleared and the appearance of any new lesions. Efficacy was evaluated by comparing the number of target lesions present at the final clinic visit with the original number counted at baseline. Investigators determined the baseline TLNS by placing a 5 [cm.sup.2] transparent grid over one to three designated treatment areas (containing a combined minimum of five AK lesions), outlining the lesions, and counting the total number of lesions present. Care was taken to ensure that the grid was realigned at each visit using facial landmarks. The CLNS consisted of the total number of lesions within the designated treatment blocks after baseline, including target lesions as well as any new lesions.
The Investigator's Global Improvement Index (IGII), was the qualitative efficacy measurement. The IGII was used to rate overall improvement at each post-baseline clinic visit. The Investigator evaluated the change in lesion status on a 7-point scale ranging from -2 to 4 points (-2 = Significantly worse; -1 = Slightly worse; 0 = No change; 1 = Slightly improved; 2 = Moderately improved; 3 = Significantly improved; 4 = Completely improved).
Digital photographs of lesions included in the designated treatment blocks were taken at all clinic visits. The confidentiality of the patient was ensured by avoiding the capture of a patient's eyes or entire face.
The tolerability of 3.0% diclofenac sodium gel was assessed by the frequency and severity of adverse events that were considered by the investigator to be related to study medication.
Patients
Patients were included in the study if they were 18 years of age or older, and had a clinical diagnosis of five or more AK lesions contained in 1 to 3 blocks (5 cm2) on the forehead, central face, or scalp. Patients who entered the study agreed to minimize use of oral NSAIDs during the treatment period. Women of childbearing potential agreed to use reliable methods of contraception for the duration of the study. Patients were excluded if they:
1. Had an allergy to aspirin, other NSAIDs, or any inactive component of 3.0% diclofenac sodium gel.
2. Were pregnant or intended to become pregnant during the course of the study, or had a stomach ulcer, stomach bleeding, severe liver or kidney disease, or other serious disease or condition that could interfere with the study evaluation also were excluded from the study.
3. Had a dermatological condition (including psoriasis) at the designated site that could affect the absorption, accumulation, and/or metabolism of 3.0% diclofenac sodium gel or could interfere with clinical assessments.
4. Had used any of these topical medications within the 2 months prior to screening: masoprocol, 5-fluorouracil, cyclosporine, retinoids, trichloroacetic acid/lactic acid/peel, and 50% glycolic acid peel.
Drug Application
The AK lesions of patients were treated twice daily with a topical application of 3.0% diclofenac sodium gel. Patients were instructed to gently smooth the gel onto the affected areas of the forehead, central face, or scalp. Patients were instructed to refrain from extensive sun exposure and the use of sun lamps, and to discuss the use of any concomitant topical drug products with their physician before applying to the affected areas.
Statistics
Change from baseline in the TLNS and CLNS was summarized by visit and for all visits using the mean, standard deviation, median, minimum, and maximum. Proportions of subjects who achieved [greater than or equal to]75% AK clearance at Days 90 and 120 (follow-up) and the corresponding 95% confidence intervals (CI) were tabulated by study site and for all sites combined. A similar analysis was conducted to derive the proportion of subjects achieving 100% AK lesion clearance and the corresponding 95% CI.
Results
Study Patients
Of the 76 patients who entered the study, 67 (88%) patients completed the study (Table 1). The reasons for discontinuation from the study were adverse events (three patients), patient consent withdrawn (three patients), lost to follow-up (one patient), and other (two patients; one patient moved to another geographic area and one patient was non-compliant with treatment). The age of the patients ranged from 48 years to 100 years, and the mean age was 68.2 years. Most patients were men (87%), and all patients were Caucasian. The BSI score indicated that AK lesions were mild (33%) or moderate (58%) for most patients. The designated treatment areas included the forehead (58%), central face (30%), and scalp (32%). Patients had Fitzpatrick skin type I (27%), II (35%), III (28%), or IV (11%); no patient had a skin type of V or VI. Approximately half (52%) of the patients used topical preparations (i.e., moisturizers, sunscreens) at the drug application site during the study. Oral NSAID or aspirin were used by 65% of the patients during the study, and approximately half (51%) of the patients used these medications once a day. The majority of patients were compliant with the medication regimen, although compliance tended to decrease over time. The percentage of patients who reported missing six or less applications ranged from 93% percent at Day 30 to 75% at Day 90.
Efficacy Evaluation
Target Lesion Number Score (TLNS)
The mean TLNS at baseline was 8.4 lesions. Treatment with 3.0% diclofenac sodium gel demonstrated a steady decrease in the TLNS (Table 2). The mean for the TLNS decreased to 1.4 lesions at Day 90 of treatment, and 0.8 lesions at Day 120 (follow-up). Although improvement was most pronounced at Days 90 and 120 (follow-up), marked decreases in AK lesions were observed as early as Day 30 (mean, 6.1 lesions) and Day 60 of treatment (mean, 3.2 lesions). The percent decrease from baseline in the TLNS is summarized in Figure 1. There was an 83% mean decrease from baseline in the TLNS at Day 90, and a 90% mean decrease from baseline at Day 120 (follow-up). Figure 2 illustrates the steady decrease over time that was observed in AK lesions at 30, 60, and 90 days of treatment with 3.0% diclofenac sodium gel, and at 120 days (follow-up).
[FIGURE 1 OMITTED]
[FIGURE 2 OMITTED]
[FIGURE 3 OMITTED]
The percentage of patients with [greater than or equal to]75% and 100% AK lesion clearance based on the TLNS is shown in Figures 3 and 4, respectively. At Day 90 of treatment, 78% (95% CI [68%, 87%]) of patients had [greater than or equal to]75% AK lesion clearance based on the TLNS. Patients continued to show improvement post-treatment with 85% (95% CI [77%, 94%]) of patients demonstrating [greater than or equal to]75% AK lesion clearance at Day 120 (follow-up). Improvement was also demonstrated by the rate of 100% AK lesion clearance. At Day 90 of treatment 41% (95% CI [29%, 52%]) of patients had 100% AK lesion clearance based on the TLNS, and patients continued to improve post-treatment with 58% (95% CI [46%, 70%]) of patients demonstrating 100% AK lesion clearance at Day 120 (follow-up).
[FIGURE 4 OMITTED]
Cumulative Lesion Number Score
Actinic keratosis lesion clearance based on the CLNS, which included target lesions as well as new lesions appearing within the designated treatment areas, demonstrated improvement that was similar to the findings for the TLNS with a steady decrease in the CLNS during the study (Table 2). The mean for the CLNS was 1.8 lesions at Day 90 of treatment, and 1.2 lesions at Day 120 (follow-up). The percent decrease from baseline (TLNS at baseline) in the CLNS is summarized in Figure 1. There was a 78% mean decrease from baseline in the CLNS at Day 90, and an 83% mean decrease from baseline at Day 120 (follow-up).
The percentage of patients with [greater than or equal to]75% and 100% lesion clearance based on the CLNS is shown in Figures 3 and 4, respectively. At Day 90 of treatment, 69% (95% CI [58%, 80%]) of patients had [greater than or equal to]75% AK lesion clearance based on the CLNS. Patients continued to show improvement post-treatment with 72% (95% CI [61%, 82%]) of patients demonstrating [greater than or equal to]75% AK lesion clearance at Day 120 (follow-up). With regard to 100% AK lesion clearance, by Day 90 of treatment 32% (95% CI [22%, 43%]) of patients had a 100% AK lesion clearance based on the CLNS, and patients continued to improve post-treatment with 45% (95% CI [33%, 57%]) of patients demonstrating 100% AK lesion clearance at Day 120 (follow-up).
Investigator Global Improvement Index
The results of the IGII indicated that most investigators considered the AK lesions of patients to be significantly improved (45%) or completely improved (27%) at Day 90 of treatment (Figure 5). At Day 120 (follow-up), the investigators continued to consider the AK lesions as significantly improved (42%) or completely improved (42%). A shift from baseline to Day 120 (follow-up) in the IGII showed greater improvement in patients with AK lesions rated as mild or moderate based on the BSI compared to patients with severe BSI. Investigators considered most patients with a mild or moderate BSI to be significantly improved (mild BSI, 26%; moderate BSI, 51%) or completely improved (mild BSI, 68%; moderate BSI, 37%) at Day 120 (follow-up). In contrast, fewer patients with severe BSI were considered by investigators to be significantly improved (29%) or completely improved (0 patients) at Day 120 (follow-up).
[FIGURE 5 OMITTED]
Safety and Tolerability Evaluation
A total of 39 patients (51%) experienced at least one adverse event related to study medication during the study. Related adverse events that occurred in two or more patients are shown in Table 3. The most common related adverse events occurred in the skin and appendages at the application site (32 patients, 42%) with the most frequently reported related adverse events ([greater than or equal to]5%) being dry skin, rash, exfoliation, and pruritus. Most related adverse events were mild or moderate in severity; pruritus (three patients) was the only related adverse event reported as severe by more than one patient. Three patients discontinued from the study due to an adverse event. Severe adverse events were experienced by 4 patients (5%); none were treatment related.
Discussion
Topical application of 3.0% diclofenac sodium gel twice daily for 90 days in this multi-center, single-arm, open-label study demonstrated clinically significant improvement in AK lesion clearance, and was generally well tolerated. After 90 days of treatment, 78% of patients had a [greater than or equal to]75% clearance of target AK lesions (TLNS). One month after completing treatment (Day 120 [follow-up]), the percentage of patients with a [greater than or equal to]75% clearance of target AK lesions had increased to 85%. Although improvement was most pronounced at Days 90 and 120 (follow-up), marked decreases in AK lesions were observed as early as Day 30 and Day 60 of treatment. In addition, complete clearance of AK lesions as assessed by the TLNS (Day 90, 41%; Day 120 [follow-up], 58%) was observed. The CLNS, which includes target lesions as well as new lesions in a designated area, also showed improvement after 90 days of treatment with topical 3.0% diclofenac sodium gel with 69% of patients showing [greater than or equal to]75% AK lesion clearance, and 72% showing [greater than or equal to]75% AK lesion clearance at Day 120 (follow-up). In addition, complete clearance of AK lesions as assessed by the CLNS (Day 90, 32%; Day 120 [follow-up], 45%) was observed. The results of the IGII confirmed the findings of the TLNS and the CLNS. At 90 days of treatment, investigators considered AK lesions to be significantly improved (45%) or completely improved (27%), and their assessment at Day 120 (follow-up) indicated that the majority of AK lesions were significantly improved (42%) or completely improved (42%). This data compares favorably with results obtained for topical 5-Fluorouracil products (15).
These results are consistent with the findings in previous double-blind, vehicle-controlled studies with topical 3.0% diclofenac sodium gel where patients demonstrated complete clearance of AK lesions based on the TLNS (3.0% diclofenac sodium gel, 33% to 50% vs. placebo, 10% to 20%) and the CLNS (3.0% diclofenac sodium gel, 31% to 47% vs. placebo, 10% to 19%) after 60-90 days of treatment. The IGII showed that investigators considered lesions completely improved in 31% to 47% of patients treated with 3.0% diclofenac sodium gel compared with 10% to 19% of patients treated with placebo (10,11). A limitation of the present study is that it is a single-arm study design, thus allowing some investigator bias. Vehicle effects are also possible. Therefore, these results should be generalized with caution.
Although there is controversy concerning the definition of AK, there is consensus that it is the result of excessive sun exposure, it can progress to SCC, and that it responds to treatment. The rationale for treatment of AK lesions is the removal of damaged epidermal cells before they progress to SCC when they need more aggressive treatments and have less favorable outcomes. The major approaches to AK therapy include surgical and physical treatments (cryosurgery, curettage and/or electrosurgery, dermabrasion, chemical peels, photodynamic therapy), and topical pharmacologic therapy with the choice of therapy being dependent on the extent of AK lesions, and the potential tolerance for side effects. patients in this study with mild or moderate BSI were considered by investigators to show more improvement in AK lesions compared with patients who had a severe BSI.
Topical pharmacologic therapy is the least invasive of these therapeutic procedures, and an alternative to other topical agents is represented by topical application of 3.0% diclofenac sodium gel. The mechanism of action of 3.0% diclofenac sodium gel is not completely understood; however, it is thought to be related to NSAID inhibition of the cyclo-oxygenase pathway and a subsequent decrease in the end products of arachidonic acid metabolism (12,13,14). Treatment with 3.0% diclofenac sodium gel in this study was shown to decrease the number of AK lesions as measured by [greater than or equal to]75% and 100% lesion clearance rates.
The majority of patients were compliant with the medication regimen. The percentage of patients who reported missing six or less applications ranged from 93% percent at Day 30 to 75% at Day 90. The high compliance is consistent with the ease of treatment with topical 3.0% diclofenac sodium gel. Topical application of 3.0% diclofenac sodium gel was well tolerated with few side effects. A total of 39 patients (51%) experienced at least one adverse event considered to be related to 3.0% diclofenac sodium gel during the study. Dry skin, rash, exfoliation, and pruritus at the application site were most commonly reported adverse events, and most of these adverse events were mild or moderate in severity. Three patients discontinued due to an adverse event; no patient experienced a treatment-related serious adverse event. The safety profile in this study is similar to the safety data reported in previous studies on topical 3.0% diclofenac sodium gel (10,11). Treatment with 3.0% diclofenac sodium gel appears to have a more favorable tolerability profile compared with other topical pharmacologic agents such as topical 5-fluorouracil that have persistent side effects, which impair compliance (15).
In conclusion, this study demonstrated the efficacy and tolerability of topical 3.0% diclofenac sodium gel in the treatment of AK lesions. Clinically significant decreases were observed in the clearance of AK lesions at rates of [greater than or equal to]75% and 100% clearance. Just as importantly, these results occur without significant morbidity or difficulty in tolerance. The topical application of 3.0% diclofenac sodium gel provides a convenient, safe, effective, and well-tolerated approach to the treatment of AK.
References
1. Salasche SJ. Epidemiology of actinic keratoses and squamous cell carcinoma. J Am Acad Dermatol 2000; I: s4-s7.
2. Frost CA, Green AC. Epidimeology of solar keratoses. Br Jr Dermatol 1994; 131(4):455-64.
3. Marks R, Rennie G, Selwood TS. Malignant transformation of solar keratoses to squamous cell carcinoma. Lancet 1988; 1(8589):795-7.
4. Mittelbronn MA. et al. Frequency of pre-existing actinic keratosis in cutaneuous squamous cell carcinoma. Int J Dermatol 1998; 37(9):677-81.
5. Lober BA, Lober CW. Actinic keratosis is squamous cell carcinoma. J Am Acad Dermatol 2000; I: s8-s10.
6. Weinstock M. Deaths from skin cancer among the elderly: epidemiological patterns. Arch Dermatol 1997; 133; 1201-9.
7. Chen JG, et al. Cost of Nonmelanoma skin cancer treatment in the United States. Dermatologic Surgery 2001; 27:(12):1035-1038.
8. Fleischer AB Jr. et al. Procedures for skin diseases performed by physicians in 1993 and 1994: analysis of data from the National Ambulatory Medical Care Survey. J Am Acad Dermatol, 1997 Nov; 37(5 Pt 1):719-24.
9. Marks R, et al. The incidence of non-melatonic skin cancer in an Australian population: results of a five-year prospective study. Med J Aust 1989; 150:475-8.
10. Wolf JE Jr. et al. Topical 3.0% diclofenac in 2.5% hyaluronan Gel in the treatment of actinic keratoses. Int J Dermatol 2001; 40:709.
11. Rivers JK, et al. Topical treatment of actinic keratoses with 3.0% diclofenac in 2.5% hyaluronan gel. Br Jr Dermatol 2002; 146:94-100.
12. Marnett LJ. Generation of mutagens during arachidonic acid metabolism. Cancer Metastasis Rev 1994; 13: 303-308.
13. Isaacs JT. Role of programmed cell death in carcinogenesis. Environ Health Perspect 1993; 101:27-33.
14. Masferrer JL. et al. Antiangiogenic and antitumor activities of cyclooxygenase-2 inhibitors. Cancer Res 2000; 60: 1306-1311.
15. Jorizzo J. et al. Randomized trial evaluating a new 0.5% fluorouracil formulation demonstrates efficacy after 1-, 2-, or 4-week treatment in patients with actinic keratosis. Cutis 2002; 70:335-339.
CHRISTOPHER NELSON MD (1), DARRELL RIGEL MD (2) STACY SMITH MD (3), NEIL SWANSON MD (4), JOHN WOLF MD (5)
1. UNIVERSITY OF IOWA, IOWA CITY, IOWA
2. NEW YORK UNIVERSITY, NEW YORK, NEW YORK
3. THERAPEUTICS CLINICAL RESEARCH, LAJOLLA, CALIFORNIA
4. OREGON HEALTH AND SCIENCE UNIVERSITY, PORTLAND, OREGON
5. BAYLOR COLLEGE OF MEDICINE, HOUSTON, TEXAS
COPYRIGHT 2004 Journal of Drugs in Dermatology, Inc.
COPYRIGHT 2005 Gale Group