Gamma-hydroxybutyrate acid (GHB) is a naturally occurring analog of GABA in the mammalian brain and can be therapeutically used for basic sedation in intensive care units. Although its application is discussed controversially, GHB is suspected to protect neuronal tissue against ischemic damage. GHB was tested for an acute effect on electrophysiologic parameters of guinea pig hippocampal tissues exposed to ischemic conditions. With application of 0.5 mM GHB, an acute protective effect was observed. The aim of the present paper is to discuss our experimental results as well as pathophysiological mechanisms of GHB and its clinical applicability. [Neural Res 2003; 25: 759-763]
Keywords: Gamma-hydroxybutyrate; GABA; ischemia; neuroprotection; hippocampal slice
INTRODUCTION
Gamma-hydroxybutyrate (GHB) was first identified and synthesized almost 40 years ago1-4. In the 1970s GHB was applied as an intravenous narcotic agent in both Europe and Japan. However, due to its suspected high incidence of vomiting and seizures when administered in narcotic doses, GHB was applied for mild sedation at low concentrations in the USA5-8. As GHB was increasingly misused as a recreational psychoactive drug which possesses a high risk for poisoning, it was banned (schedule I = no medical use, high potential for abuse) in the United States in 1991. It is still permitted for strictly medical use in many European countries9-11. Although its application is discussed controversially GHB is suspected to protect neuronal tissue against ischemic damage12.
The pathophysiological mechanisms of GHB, the experimental data according its cerebroprotective potential, and its clinical applicability are critically reviewed and discussed.
Occurrence and distribution
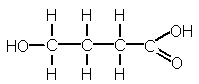
Gamma-hydroxybutyrate (C4H803) is a short-chain fatty acid, which is a naturally occurring analogue of gamma-aminobutyric acid (GABA) in the mammalian brain
It is derived primarily following GABA metabolism via GABA-transaminase and succinic semialdehyde reductase13,14. GHB appears heterogeneously distributed in the brain15 and has been proposed to play a role as a inhibitory neurotransmitter16. Neuromodulatory functions are regulated via high and low affinity binding sites of specific receptors17,18 which are found mainly in the rostral part of the brain (frontal cortex, hippocampus, striatum, thalamus, dopaminergic nuclei). These receptors were also present in neuronal cell cultures19,20. In mammalian brains GHB tissue concentration is highest in human and monkey brain, reaching about 11-25 [mu]M in the striatum. Only in developing brains has the concentration of tissue GHB been found to be higher17,21. GHB also occurs in kidney, heart, skeletal muscles and brown fat tissues22.
Second messenger system
After administration of 400 mg kg^sup -1^ body-weight (BW), GHB, absolute levels of cyclic guanosine monophosphate (cGMP) have been found to increase two-fold in the rat hippocampus, whereas the cyclic adenosin monophosphate (cAMP) level remained unchanged. These doses seem to induce local depolarization which increases the local concentration of neuronal Ca^sup 2+^ and cGMP. Raising the GHB doses above 500mg kg^sup -1^ BW abolishes the cGMP increase23. It is postulated that high GHB concentrations most probably develop a GABAergic influence, which counteracts the cGMP increase17,24.
Dopaminergic system
Whether GHB increases or decreases synaptic dopaminergic activity and/or release depends partly on the dose, the route of administration, the time at which levels are measured, and the presence or absence of general anesthesia6,25. Systemic administration of low doses of GHB (30-300 mg kg^sup -1^ BW) in the rat induces endogenous GHB levels up to 250 [mu]M in the brain. This concentration inhibits burst activity and modulates firing of the dopaminergic terminals26-28. The same effects were observed after local administration of GHB In the substantia nigra. A decrease in dopamine release in the striatum following low doses of GHB is mimicked by GABA^sub B^ agonists and reduced by GABA^sub B^ blockers17,27. Structural analogues of GHB, possessing better affinity for binding sites than GHB itself, can replace GHB at lower doses29. With increased GHB concentration, the initial decline of the dopamine release is followed rapidly by a tissue accumulation of dopamine in the rat forebrain17,30. One probable mechanism for this accumulation might be an escape of dopamine synthesis from inhibitory control on presynaptic dopamine receptors31. It seems that the GHBergic system influences the control of the dopaminergic system mainly by a change of impulse flow in the nigro-striatal and meso-corticolimbic pathways17,27.
Serotonergic system
Additionally, in vivo and in vitro experiments demonstrate an influence of the GHB system on the serotonergic activity in the brain. It is still unclear whether this influence is caused directly or indirectly via interaction with other systems (dopaminergic, GABA-ergic). Higher doses of GHB (400-500 mg kg^sup -1^ BW) in rats induce an increase of the serotonin turnover in striatum and in the meso-limbic areas17,32. This GHB-induced stimulation of the tissue serotonin turnover is most probably due to an increase in the transport of the serotonin precursor, tryphtophan, to the brain and its uptake by serotonergic cells17,33. As the serotonergic system is involved in the regulation of sleep, mood and anxiety, the stimulation of this system by GHB may be responsible for the neuropharmacological effects of GHB.
Other systems
GHB, administered in high doses (400-700 mg kg^sub -1^) in rats, increases the release of endogenous opiates like donorphin and beta-endorphin16,34. Morphin-like effects of GHB can be partially reversed by the opiate antagonist naloxone. Feigenbaum and Simantov35, who examined the effect of different concentrations of GHB on mu, delta, and kappa-opioid receptor bindings demonstrated that GHB does not bind to these receptors. Consequently, GHB seems not to be a direct opiate receptor agonist.
GHB stimulates pituitary growth hormone (GH) release up to nine-fold after 30 and up to sixteen-fold after 60 min after administration of 2.5 g in healthy men36. The mechanism by which GHB stimulates the GH is still unknown.
Pharmakodynamic
GHB is usually administered in the form of a chemical salt (Na-GHB, C4H8NaO3) which freely penetrates the blood-brain barrier4,14. GHB possesses a very short elimination half-life of about 30 min37. The GHB blood plasma concentration amounts to 200-300 [mu]g ml^sup -1^ following an application of 60 mg kg^sup -1^ BW in men38. A steady state blood plasma concentration can be achieved by a bolus injection of 40 mg kg^sup -1^ BW with a following continuous injection of 10-15 mg kg^sup -1^ BWh^sup -1^ 39. GHB is completely metabolized in the liver to the natural substrates carbon dioxide and water without accumulation in central or peripheral tissues. Clinical effects of GHB like sedation can be partly antagonized by physostigminsalicylate, which is a functional GHB antagonist5,39. After administration of GHB at doses of 40-50 mg kg^sup -1^ BW, the arterial blood pressure and the heart frequency remain stable or react with a slight decrease (
Cerebroprotection
A number of observations indicate that GHB reduces energy substrate consumption in both brain and peripheral tissues and that it can protect neuronal tissues from ischemic damage42. One of the main findings is a reduction in cerebral glucose consumption after GHB application in rats with an unchanged cerebral metabolic rate of oxygen and cerebral blood flow12,43. Furthermore, direct radical scavenger effects of GHB are also discussed42. Positive effects of GHB on the reduction of intracranial pressure after severe head trauma as well as on the Glasgow Coma Scale are published41,44. Until now no class 1 data exist about this topic.
MATERIALS AND RESULTS
Ischemic tolerance
It is speculated that GHB by reduction of cerebral metabolism could potentially be a cerebroprotective agent12. The aim of our experiments was to analyze the acute effect of GHB on bioelectrical changes in hippocampal brain slices exposed to ischemia. Ischemic conditions were mimicked by deprivation of oxygen and lowering of glucose. Under critical ischemic conditions, the occurrence of a sudden negative shift of the direct current (DC) potential, the so-called anoxic depolarization (AD) is brought about by a synchronized depolarization of neurons. This sudden depolarization is accompanied by an excessive intracellular influx of calcium and sodium-ions, a release of glutamate, an increase in extracellular potassium concentrations (up to 60 mmol l^sup -1^), and a reduction of the extracellular volume fraction. It represents the endpoint of energy depletion of neuronal tissue during persistent ischemia in vivo and in vitro45,46.
Adult guinea pigs (n = 60) weighing between 340 and 440 g were anesthetized with the short acting barbiturate methohexital (250 mg kg^sup -1^ i.p.). The brain was rapidly removed and ice-cooled in artificial cerebrospinal fluid (aCSF). The aCSF contained (in mmol l^sup -1^): NaCl 124, KCl 4, NaH^sub 2^PO^sub 4^ 1.24, MgSO^sub 4^ 1.3, NaHCO^sub 3^ 26, glucose 10 and CaCl^sub 2^ 2. The hippocampus was dissected and cut transversely in 500 [mu]m slices (n = 72). After a pre-incubation period, slices were transferred into the recording chamber (34[degrees]C, pH 7.4) and placed at the interface of aCSF and humidified gas (95% O2 and 5% CO2). GHB was added to the aCSF at different concentrations (0.25, 0.5, 1, 5, 10 mmol l^sup -1^). Ischemic conditions were established by replacing N^sub 2^ for O2 in an interface chamber, and by lowering the glucose concentration (10 to 2 mmol l^sup -1^).
A bipolar stimulation electrode (0.2 msec, 0.1 Hz) was placed in the stratum radiatum between CA2 and CA3 areas of the hipocampus. The local direct current (DC) potential and the evoked potentials (EP) were recorded in the CA1 region.
Ischemic episodes were induced and terminated when the anoxic depolarization (AD) of the DC potential had reached its peak. Criterion of an increasing ischemic tolerance was a prolongation of the AD latency. Statistical analysis was performed using Mann-Whitney Rank Sum test.
A significant extension (p
The threshold for GABA^sub B^ receptor activation by GHB was found between 0.5 and 1 mM GHB8,47. As mentioned above such doses of GHB are known to induce a decrease in dopaminergic firing and a reduction of dopamine release6,28,34,48,49. The inhibition of neuronal activity by blocking the synaptic transmission consequently leads to a reduction of cerebral energy consumption. The reduction of the synaptic activity and therewith a lowered consumption of the high energy phosphates (ATP) would preserve the Na^sup +^/ K^sup +^-pump function during ischemia. Furthermore the membrane potential can be maintained longer and AD should be expected to be delayed as demonstrated in our experiments.
ELECTRO-ENCEPHALOGRAM
Peripherally, administration of clinical doses of GHB induces a modification of the EEC pattern (electro-encephalogram) in humans and most other mammals. After administration of 40-60 mg kg^sup -1^ BW GHB, physiological alpha/beta wave activity changes to a delta and theta rhythm39,50. Higher doses of GHB (150-300 mg kg^sup -1^ BW) induce modifications in EEG pattern, which resemble absence like seizures (3 cycles sec^sup -1^ spike and waves). In rodents, GHB doses sufficient to increase GHB level to about 240 [mu]M (250-400 mg kg^sup -1^ BW) induce EEC hypersynchrony and bursts of 4-6 cycles sec^sup -1^ with behavioral arrest15. These EEG changes are the origin of the 'GHB model of petit mal epilepsy'51. EEC profile can be antagonized by anti-petit mal drugs such as valproate or ethosuximide52. It might be proposed that the GHB-induced GABAergic modulations are the basis for the induction and maintenance of seizures in the GHB model. However, within the clinical range of GHB application (40-60 mg kg^sup -1^ BW) Entholzner et al.10 did not find any seizure like patterns in EEG.
SLEEP AND SEDATION
GHB in high doses was used almost 35 years ago in Europe and Japan as an additive in general anesthesia, due to its marked hypnotic action7,8,54. With doses in a range of 35-90 mg kg^sup -1^ BW i.v., GHB induces sedation. Narcotic effects are produced by increasing GHB concentrations40,55. GHB-induced sleep seems to be a physiological resemblance to normal sleep by improving the REM (rapid eye movement) efficiency and increasing its latency. GHB given to narcoleptic patients during the night facilitates sleep reconstruction due to an improvement of both non-REM and REM sleep. In patients with narcolepsy, a decreased cataplexy and an improved nocturnal sleep quality were demonstrated56. Eight hours after intake, GHB disappears from the blood plasma. It was demonstrated that the sedative properties of GHB could be counteracted by the GHB receptor antagonist NCS-382. It seems that GHB receptors are implicated directly in the sleep-inducing properties of GHB57,58.
During normal and GHB sleep, the CNS continues to be responsive to noxious stimuli like e.g. pain, a factor which sets limits on the isolated use of GHB in anesthesia8. On the other hand GHB increases the release of endogenous opiates like donorphin and beta-endorphin which may lead to a potentiation of additional administerd opioids16,31.
ABUSE
Despite many therapeutic and economic properties like good sedation, neuroprotective effects, cardiovascular stability and a low priced production, GHB is banned in the United States since 1991. The main reason for this ban is the use of GHB as a recreational drug, sold on streets as 'Liquid Ecstasy', 'Liquid X', 'Salty Water' and 'Fantasy'. In 1991 the Centers for Disease Control reported 57 cases of GHB poisoning. In the following years GHB poisoning increased dramatically, especially in the United States and Australia10. GHB is available as a colorless, odorless liquid, powder or capsules. Improper manufacture can lead to extremely toxic substances. Especially, the synergy of GHB and alcohol or other recreational drugs is of clinical concern56,59. Tolerance and physical dependence after prolonged use of high doses have been reported, resulting in withdrawal symptoms like muscle cramps, tremors and anxiety60. During sleep an increase in growth hormone secretion was observed, which has led to the unsubstantiated use of GHB by body-builders56.
Signs of poisoning have been reported in variable doses with a range of 2.5 g to 30 g. Adverse effects are vomiting, confusion, delirium, bradycardia, Cheyne-Stokes breathing and coma resulting in death. The adverse effects are seen after 15 min of oral ingestion. Naloxon administration in the treatment of GHB coma is controversial. One therapeutic option is physiostigmin, which provides an accelerated awakening56. Recovery occurs within 7 h due to the short half-life of the drug. On the other hand several studies have indicated that exogenous GHB could reduce voluntary ethanol intake in alcohol-preferring rats. GHB also suppresses ethanol withdrawal symptoms, both in ethanol-dependent rodents and humans61.
CONCLUSION
GHB has some properties which make it attractive for therapeutic use. Advantages of GHB are a good sedation with reduced cardiac and respiratory depression, low costs and an absence of irritation of veins. In some European and Japanese departments, GHB is administered as an alternative to established sedative agents, such as benzodiazepine, especially in stroke patients and patients having received a cardiac bypass39,62. Our experimental results demonstrate an acute prolongation of ischemic tolerance in vitro with 0.5 mM GHB. Unfortunately, the therapeutic range of GHB In this model is relatively small, due to some U-shaped effects. Despite its misuse as a recreational drug, GHB seems to be an alternative to benzodiazepines, especially when cerebral ischemia is suspected. Randomized double-blind studies have to be conducted to investigate its cerebroprotective effect.
ACKNOWLEDGEMENTS
With support of the Deutsche Forschungsgemeinschaft (AZ:Wa 604/2-1).
REFERENCES
1 Bessmann SP, Fishbein WN. Gamma-hydroxybutyrate, a normal brain metabolite. Nature 1963; 200: 1207-1208
2 Fishbein WN, Bessmann SP. [gamma]-Hydroxybutyrate in mammalian brain. J Biol Chem 1961; 239: 357-361
3 Roth RH. Formation and regional distribution of [gamma]-hydroxybutyric acid in mammalian brain. Biochem Pharmacol 1970; 19: 3013-3019
4 Roth RH, Giarman NJ. Natural occurrence of gamma-hydroxybutyrate in mammalian brain. Biochem Pharmacol 1970; 19: 1087-1093
5 Henderson RS, Holmes CM. Reversal of the anaesthetic action of sodium gamma-hydroxybutyrate. Anaesth Intensive Care 1976; 4: 351-354
6 Howard SG, Feigenbaum JJ. Effects of [gamma]-hydroxybutyrate on central dopamine release in vivo. Biochem Pharmacol 1996; 53: 103-110
7 Laborit H. Sodium-4-hydroxybutyrate. Int J Neuropharmacol 1964; 3: 433-452
8 Vickers MD. Gamma hydroxybutyric acid. Clinical pharmacology and current status. Proc R Soc Med 1968; 61: 821-824
9 Centers for Disease Control. Gamma hydroxybutyrate use in New York and Texas, 1995-1996. J Am Med Assoc 1997; 277: 1511
10 Centers for Disease Control. Multistate outbreak of poisonings associated with the illicit use of gamma hydroxybutyrate. J Am Med Assoc 1991; 265: 447-448
11 Stell JM, Ryan JM. Ecstasy and neurodegeneration. Gamma-hydroxybutyrate is a new recreational drug that may lead to loss of consciousness. BMJ 1996; 312: 1493-1494
12 Kuschinsky W, Suda S, Sokoloff L. Influence of gamma-hydroxybutyrate on the relationship between local cerebral glucose utilization and local cerebral blood flow in the rat brain. J Cereb Blood Flow Metab 1985; 5: 58-64
13 Rumigny JF, Cash C, Mandel P, Vincendon G, Maitre M. Evidence that specific succinic semialdehyde reductase is responsible for [gamma]-hydroxybutyrate synthesis in brain tissue slices. FEBS Lett 1981; 134: 96-98
14 Xie X, Smart TG. [gamma]-Hydroxybutyrate hyperpolarizes hippocampal neurones by activating GABA^sub B^ receptors. Eur J Pharmacol 1992; 212: 291-294
15 Snead OC III. The gamma-hydroxybutyrate model of absence seizures: Correlation of regional brain levels of gamma-hydroxybutyric acid and gamma-butyrolactone with spike wave discharges. Neuropharmacology 1991; 30: 161-167
16 Vayer P, Mandel P, Maitre M. Gamma-hydroxybutyrate, a possible neurotransmitter. Life Sci 1987; 41: 1547-1557
17 Maitre M. The gamma-hydroxybutyrate signaling system in brain: Organization and functional implications. Prog Neurobiol 1997; 51: 337-361
18 Snead OC III, Nichols AC. Gamma-hydroxybutyric acid binding sites: Evidence for coupling to a chloride anion channel. Neumpharmacology 1987; 26: 1519-1523
19 Kemmel V, Taleb O, Perard A, Andriamampandry C, Siffert JC, Mark J, Maitre M. Neurochemical and electrophysiological evidence for the existence of a functional gamma-hydroxybutyrate System in NBC-20 neurons. Neuroscience 1998; 86: 989-1000
20 Ratomponirina C, Mode Y, Hechler V, Maitre M. Gamma-hydroxybutyrate receptor binding in rat brain is inhibited by guanyl nucleotides and pertussis toxin. Neurosci Lett 1995; 189: 51-53
21 Snead OC III, Morley BJ. Ontogeny of gamma-hydroxybutyric acid. I. Regional concentration in developing rat, monkey and human brain. Brain Res 1981; 227: 578-589
22 Chin MY, Kreutzer RA, Dyer JE. Acute poisoning from gamma-hydroxybutyrate in California. West J Med 1992; 156: 380-384
23 Vayer P, Gobaille S, Mandel P, Maitre M. 3'-5' cyclic-guanosine monophosphate increase in rat brain hippocampus after gamma-hydroxybutyrate administration. Prevention by valproate and naloxone. Life Sci 1987; 41: 605-610
24 Vayer P, Maitre M. Gamma-hydroxybutyrate stimulation of the formation of cyclic GMP and inositol phosphates in rat hippocampal slices. J Neurochem 1989; 52: 1382-1387
25 Madden TE, Johnson SW. Gamma-hydroxybutyrate is a GABA^sub B^ receptor agonist that increases a potassium conductance in rat ventral tegmental dopamine neurons. J Pharmacol Exp Ther 1998; 287: 261-265
26 Anden NE, Stock G. Inhibitory effect of gammahydroxybutyric acid and gammaaminobutyric acid on the dopamine cells in the substantia nigra. Naunyn Schmiedebergs Arch Pharmacol 1973; 279: 89-92
27 Nissbrandt H, Elverfors A, Engberg G. Pharmacologically induced cessation of burst activity in nigral dopamine neurons: Significance for the terminal dopamine efflux. Synapse 1994; 17: 217-224
28 Roth RH, Doherty JD, Walters JR. Gamma-hydroxybutyrate: A role in the regulation of central dopaminergic neurons. Brain Res 1980; 189: 556-560
29 Hechler V, Peter P, Gobaille S, Bourguignon JJ, Schmitt M, Ehrhardt JD, Mark J, Maitre M. Gamma-hydroxybutyrate ligands possess antidopaminergic and neuroleptic-like activities. J Pharmsco Exp Ther 1993; 264: 1406-1414
30 Dyck LE, Kazakoff CW. Acceleration of the biosynthesis of rat striatal dopamine by incubation and by administration of gamma-butyrolactone. J Neurosci Res 1982; 8: 57-65
31 Handforth A, Sourkes TL. Inhibition by dopamine agonists of dopamine accumulation following gamma-hydroxybutyrate treatment. Eur J Pharmacol 1975; 34: 311-319
32 Miguez I, Aldegunde M, Duran R, Veira JA. Effect of low doses of gamma-hydroxybutyric acid on serotonin, noradrenaline, and dopamine concentrations in rat brain areas. Neurochem Res 1988; 13: 531-533
33 Gobaille S, Schleef C, Hechler V, Viry S, Aunis D, Maitre M. Gamma-hydroxybutyrate increases tryptophan availability and potentiates serotonin turnover in rat brain. Life Sci 2002; 70: 2101-2112
34 Hechler V, Gobaille S, Bourguignon JJ, Maitre M. Extracellular events induced by gamma-hydroxybutyrate in striatum: A microdialysis study. J Neurochem 1991; 56: 938-944
35 Feigenbaum JJ, Simantov R. Lack of effect of gamma-hydroxybutyrate on mu, delta and kappa opioid receptor binding. Neurosci Lett 1996; 212: 5-8
36 Takahara J, Yunoki S, Yakushiji W, Yamauchi J, Yamane Y. Stimulatory effects of gamma-hydroxybutyric acid on growth hormone and prolactin release in humans. J Clin Endocrinol Metab 1977; 44: 1014-1017
37 Ferrara SD, Zotti S, Tedeschi L, Frison G, Castagna F, Gallimberti L, Gessa GL, Palatini P. Pharmacokinetics of gamma-hydroxybutyric acid in alcohol dependent patients after single and repeated oral doses. Br J Clin Pharmacol 1992; 34: 231-235
38 Vree TB, Damsma J, van der Borgert AG, van der Kleijn E. Pharmacokinetics of 4-hydroxybutyric acid in man, rhesus monkey and dogs. In: Frey R, ed. Neue Untersuchungen mit Gamma-Hydroxy-Buttersaure. Anaesthesiologie und Intensivmedizin, Berlin: Springer Verlag, 1978: pp. 21-28
39 Kleinschmidt S, Mertzlufft F. Gamma-hydroxybutyric acid - significance for anesthesia and intensive care medicine? Anasthesiol Intensivmed Notfallmed Schmerzther 1995; 30: 393-402
40 Pichlmeier R, Schneck H. Gamma-Hydroxybuttersaure (GHB) als Basis-Sedativum in der Intensivmedizin. Intensiv Notfallbehandl 1991; 16: 106-112
41 Plangger C. Effect of gammahydroxybutyrate on intracranial pressure, mean systemic arterial pressure and cerebral perfusion pressure in experimentally induced brain oedema of the rat. Zentralbl Neumchir 1990; 51: 24-33
42 Mamelak M. Gammahydroxybutyrate: An endogenous regulator of energy metabolism. Neurosci Biobehav Rev 1989; 13: 187-198
43 Haller C, Mende M, Schuler F, Schuh R, Schrock H, Kuschinsky W. Effect of gamma-hydroxybutyrate on local and global glucose metabolism in the anesthetized cat brain. J Cereb Blood Flow Metab 1990; 10: 493-498
44 Strong AJ. Gamma-hydroxybutyric acid and intracranial pressure. Lancet 1984; 1: 1304
45 Balestrino M. Pathophysiology of anoxic depolarization: New findings and a working hypothesis. J Neurosci Methods 1995; 59: 99-103
46 Caspers H, Speckmann EJ. Cortical DC-shifts associated with changes of gas tension in blood and tissue. In: Remond A, ed. Handbook of Electroencephalography and Clinical Neurophysiology, Amsterdam: Elsevier, 1974: pp. 41-65
47 Laborit H, Jovany J, Gerard J, Fabiani F. Resumee d'une etude experimentale et clinique sur un substrat metabolique a action centrale inhibitrice: Le 4-hydroxybutyrate de Na+. Presse Med 1960; 50: 1867-1869
48 Cash CD, Gobaille S, Kemmel V, Andriamampanclry C, Maitre M. Gamma-hydroxybutyrate receptor function studied by the modulation of nitric oxide synthase activity in rat frontal cortex punches. Niochem Pharmacol 1999; 58: 1815-1819
49 Jensen K, Mody I. GHB depresses fast excitatory and inhibitory synaptic transmission via GABA^sub B^ receptors in mouse neocortical neurons. Cerebral Cortex 2001; 11: 424-429
50 Roquefeuil B, Escuret E, Duboin MP, Baldy-Moulinier M. The effect of gamma-OH on cerebral metabolism and blood flow in human patients. Rev Electroencephalogr Neurophysiol Clin 1977; 7: 93-97
51 Snead OC III. Gamma-hydroxybutyrate model of generalized absence seizures: Further characterization and comparison with other absence models. Epilepsia 1988; 29: 361-368
52 Maitre M, Hechler V, Vayer P, Gobaille S, Cash CD, Schmitt M, Bourguignon JJ. A specific gamma-hydroxybutyrate receptor ligand possesses both antagonistic and anticonvulsant properties. J Pharmacol Exp Ther 1990; 255: 657-663
53 Entholzner E, Mielke L, Pichimeier R, Weber F, Schneck H. EEG changes during sedation with gamma-hydroxybutyric acid. Anaesthesist 1995; 44: 345-350
54 Hunter AS, Long WJ, Ryrie CG. An evaluation of gamma-hydroxybutyric acid in paediatric practice. Br J Anaesth 1971; 43: 620-628
55 Hoes MJ, Vree TB, Gueleu PJ. Gamma-hydroxybutyric acid as hypnotic. Clinical and pharmacokinetic evaluation of gamma-hydroxybutiric acid as hypnotic in man. Encephale 1980; 6: 93-99
56 Okun MS, Boothby LA, Bartfield RB, Doering PL. GHB: An important pharmacologic and clinical update. J Pharm Pharm Sci 2001; 4: 165-175
57 Mitler MM, Hajdukovic R. Relative efficacy of drugs for the treatment of sleepiness in narcolepsy. Sleep 1991; 14: 218-220
58 Schmidt C, Gobaille S, Hechler V, Schmitt M, Bourguignon JJ, Maitre M. Anti-sedative and anti-cataleptic properties of NCS-382, a gamma-hydroxybutyrate receptor antagonist. Eur J Pharmacol 1991; 203: 393-397
59 Kam PC, Yoong FF. Gamma-hydroxybutyric acid: An emerging recreational drug. Anaesthesia 1998; 53: 1195-1198
60 Galloway GP, Frederick SL, Staggers FE Jr, Gonzales M, Stalcup SA, Smith DE. Gamma-hydroxybutyrate: An emerging drug of abuse that causes physical dependence. Addiction 1997; 92: 89-96
61 Gallimberti L, Cibin M, Pagnin P, Sabbion R, Pani PP, Pirastu R, Ferrara SD, Cessa GL. Gamma-hydroxybutyric acid for treatment of opiate withdrawal syndrome. Neuropsychopharmacology 1993; 9: 77-81
62 Ueki Y. Effects of gamma-dehydroxybutyrate on monoamine metabolism and protein synthesis after transient global cerebral ischemia. No Shinkei Geka 1992; 20: 937-946
Christoph Greiner, Jens-Eric Rohl*, Ali-Gorji*, Hansdetlef Wassmann and Erwin-Josef Speckmann*
Klinik und Poliklinik fur Neurochirurgie, Universitatsklinikum Munster, Munster * Institut fur Physiologie, Westfalische Wilhelms-Universitat Munster, Munster, Germany
Correspondence and reprint requests to: Dr Christoph Greiner, Klinik und Poliklinik fur Neurochirurgie, Universitatsklinikum Munster, Albert-Schweitzer-Str. 33, 48149 Munster, Germany.
[greiner@uni-muenster.de] Accepted for publication May 2003.
Copyright Forefront Publishing Group Oct 2003
Provided by ProQuest Information and Learning Company. All rights Reserved