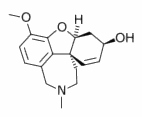
Galantamine
Galantamine (trade name Razadyne®, Reminyl®) is a medication used in the treatment of Alzheimer's disease. more...
Galantamine is mostly used as hydrobromide salt. It is a centrally acting reversible cholinesterase inhibitor in the same class as donepezil. Galantamine has a much shorter half life than donepezil (there is a newer prolonged release version available in 2004) and it also has more nicotinic receptor activity and is potentially more stimulating but also probably because of this activity tends to cause worse gastrointestinal side effects.
Total synthesis
The natural source of galantamine are certain species of daffodil and because these species are scarce and because the isolation of galanthamine from daffodil is expensive (a 1996 figure specifies 50,000 US dollar per kilogram) alternative synthetic sources are under development by means of total synthesis. One recent publication details the enantioselective organic synthesis of galanthamine and also that of morphine from a single precursor .
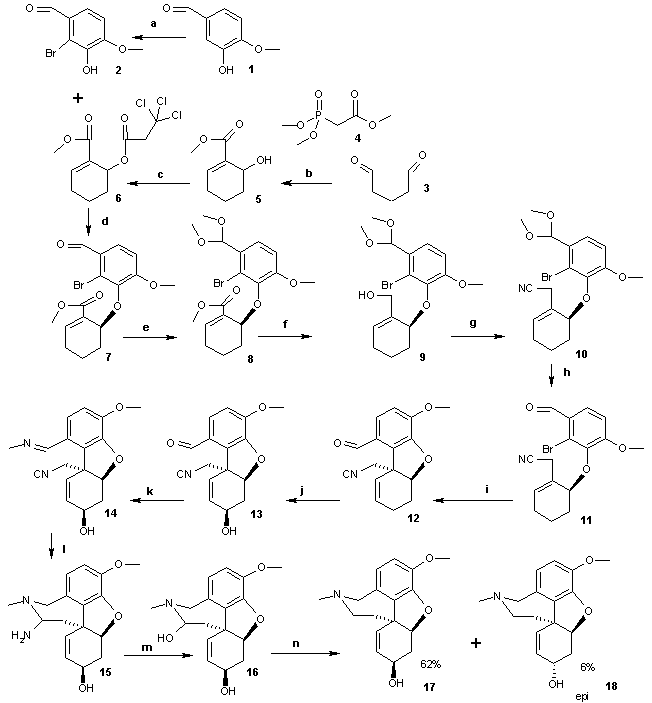
The total synthesis of galanthamine (Trost 2005) is described as follows (see scheme 1): the sequence starts by bromination by electrophilic aromatic substitution of isovanillin 1 to bromophenol 2, then by synthesis of the second intermediate 5 by reacting dialdehyde 3 in a coupled aldol reaction and Horner-Wadsworth-Emmons reaction with trimethyl phosphonoacetate 4. The hydroxyl group is activated as a leaving group by acetylation with trichloroethyl carbonate (Troc) to 6. Next an enantioselective Trost AAA reaction takes place between bromophenol 2 and carbonate 6 to the allyl ether 7. Next the aldehyde group is protected as an acetal in 8 and this step enables the to organic reduction of the ester group to the alcohol 9 with DIBAH and subsequent conversion of this alcohol to a nitrile by nucleophilic displacement to 10 followed by aldehyde deprotection to 11. The intramolecular Heck reaction to 12 creates the dihydrofuran ring. Allylic oxidation by selenium dioxide provides allylic alcohol 13 with the correct stereochemistry. The aldehyde reacts with methylamine to the imine 14 and reduction of the imine and nitrile by DIBAL-H leading to ring-closure to the hemi-aminal 15 (not isolated) followed by acid quenching gives the alcohol 16. In the final step this alcohol group is reduced to give Galanthamine 17 together with 6% of the epi isomer 18.
Read more at Wikipedia.org