Harry Howard almost made the biggest blunder of his life when he tossed the building blocks of a wonder drug into a laboratory trash bag 14 years ago. He had just created a new molecule at Pfizer's research building in Groton, Conn.-a molecule that had never existed before and that, he hoped, might eventually prove useful in treating schizophrenia. After running a test on it, Howard poured the contents of a test tube through a filter and into a flask. Then he threw out the filter and examined the liquid left behind. It didn't seem to have any of the properties he had expected. Oh well, he thought, chemicals can do weird things.
The next day, however, it occurred to him that perhaps he was looking in the wrong place. "It's a good thing I hadn't taken the trash out," he says, "because my compound was stuck in that filter."
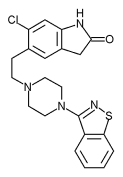
Over the course of the next decade, Pfizer would invest hundreds of millions of dollars in what Howard yanked from the trash. Today, his compound is called Geodon. It went on the market almost two years ago and, by most accounts, it's one of the best medications available to people with schizophrenia. Pfizer won't say exactly how much money it poured into Geodon, but one estimate says a successful drug can require an investment of $800 million before the Food and Drug Administration will approve it. There are thousands of failures for every success-and somehow Pfizer must pay for them as well.
In a good year, Pfizer might win approval for one or two brand-new products. It then has only a short time to earn back what it has invested. Geodon's patent, for instance, expires in March 2007, at which point generic copies will begin to flood the pharmacy shelves. By then, Pfizer will have had just six years to pay for the huge costs that went into making Geodon. The company estimates that it spends $100 million on research and development per week. Within the next five years, it will lose the patents for four of its eight most popular drugs.
Everybody loves wonder drugs, but nobody seems to want to pay for them. One industry-sponsored poll revealed that 90 percent of Americans believe the price of prescription drugs is a "major problem" and 60 percent support outright price controls. Few people appreciate that innovation doesn't come cheap, especially when drugs must pass through expensive regulatory hurdles meant to test their safety and effectiveness. Wonder drugs may seem like miracles, but they don't rain down upon us like manna. Producing even one requires a monumental effort.
Perhaps nothing can illustrate this point better than a story, and Geodon's story begins with the problem of schizophrenia. "It's one of the worst mental diseases out there," says Steven Romano, a Pfizer psychiatrist. Schizophrenics suffer from hallucinations, delusions, and social withdrawal, among other symptoms. Some 2 million Americans have the disease, and many of them can't hold down a regular job: John Nash, the Princeton mathematician portrayed by Russell Crowe in the movie A Beautiful Mind, may be this country's best-known schizophrenic. "It has exacerbations and remissions, but it's chronic-it's always there, and there's no cure," says Romano.
The earliest drugs for schizophrenia could control the hallucinations and delusions, but only at the cost of grisly side effects, such as severe muscular tremors. During the 1970s and 1980s, scientists began looking for alternatives, and Geodon had its origins in this broad effort. By the early 1990s, new products that didn't make muscles go haywire were on the market, but these had their own downsides, such as sharp weight gain. As a result, many schizophrenics were getting their main symptoms under control, only to quit their meds because they didn't like the side effects.
Harry Howard was one of several Pfizer employees in the 1980s charged with producing compounds that might improve the treatment of schizophrenics. "Discovery" scientists like Howard spend their days assembling molecules that have never existed before and then seeing how they behave in test tubes. "This part of the process is all about creativity," says Stevin Zorn, who led much of the Geodon research. "There's no cookbook on how to make a successful compound. It's art as much as science."
It's also drudgery. Even after Howard had constructed the original Geodon molecule-code-named CP88059-his creation faced long odds. "For every 150,000 compounds our scientists create, we figure one of them will make it to the marketplace," says Derek Leishman, a pharmacologist with Pfizer in England. It's so rare for a compound to make it to the shelves, in fact, that many discovery scientists will spend their entire careers working on compounds that never become approved drugs.
When a compound looks promising in the test tubes, Pfizer scientists begin putting it into animals-mostly mice, rats, and dogs. They want to make sure their proto-drug isn't toxic. They're also looking for clues about how it might act in humans. With CP88059, scientists were hoping to spot muscle rigidity, a negative side effect that had been associated with other successful antipsychotic medications. But they found nothing of the sort, which suggested that CP88059 might be a different breed of drug. By this point, in 1989, it was time for Geodon to "get canned."
This is an important milestone, as a compound moves from one side of the R&D ampersand to the other. It's called "getting canned" because the company puts out a "Candidate Alert Notice" (CAN)-basically an all- points bulletin announcing that the drug has cleared its first set of obstacles. Yet the most challenging phase still lies ahead: Fewer than 10 percent of canned compounds ever make it to the marketplace.
The discovery scientists had produced a fine, white powder that looked similar to flour. But one of the great predicaments companies like Pfizer face consists in moving from the pure science of compound creation to the applied science of developing a form of the drug that patients can use, such as a pill or capsule. The final product must be able to survive on the shelf for a couple of years, remain stable in hot and cold temperatures, and deliver enough punch in a single dose that patients won't have to pop pills a dozen times a day.
This step is harder than it sounds, but it's essential to the pharmaceutical business-and it's in this area that Pfizer made its first great contribution to humanity. In 1928, Alexander Fleming found that a common airborne mold known as penicillium could vanquish pneumonia and scarlet fever-but only in a petri dish. He had enormous trouble preserving the mold's active substance. It wasn't until the Second World War that anybody was able to produce penicillin in a useful form. Pfizer was the company that figured out how to do it.
Pfizer faced this same challenge with Geodon-converting a known substance into a product people could actually use. "It took us two years to learn how to get it into patients, and then a few more years of optimizing," says Bijan Rasadi, one of Pfizer's chief pharmaceutical scientists. The entire development process took nine years.
A canned compound eventually graduates to clinical testing, when it's put into humans for the first time. The first phase of Geodon's clinical trials began in 1991 and, as first phases always do, it involved a few dozen healthy subjects-non-schizophrenics who were willing to serve as human guinea pigs. The point of these initial tests isn't to see whether a drug works, but to determine whether it's safe for human consumption. "Geodon's first phase was uneventful," says Rachel Swift, Pfizer's executive director of clinical development. That was precisely the news she wanted to hear.
Once a drug's safety has been established in Phase One, scientists next examine its efficacy. These Phase Two trials typically involve several hundred people affected by the problem the drug is supposed to address. Researchers are generally confident that their drug will have some effect, but they need to determine ideal dosage levels.
Geodon's Phase Two trials were also uneventful-perhaps a bit too much so. Pfizer's R&D scientists saw that Geodon was helping patients, but it didn't outperform existing medications. If Geodon were ever to make it to the marketplace, it would require yet more investment, and at the end of the process there still had to be a reasonable hope that it would generate revenue. A redundant drug wouldn't. There was a strong case for putting the brakes on Geodon. This is a fairly common occurrence even for drugs that enter the second phase of clinical testing.
Yet Pfizer was also hearing from doctors involved in the trials that compliance was up. Geodon patients weren't putting on the kind of weight associated with other treatments. If this drug were able to delete that side effect, the researchers thought, then perhaps they really had something.
At the end of Phase Two trials, the white lab coats sit down with the suits, which is to say the development team presents the results to the company management (who are generally scientists themselves), and they debate whether to move forward. The third and final phase of clinical trials is incredibly expensive, so the decision at this stage is momentous.
Geodon got the green light. If it succeeded during Phase Three, the FDA would approve it and doctors would be allowed to prescribe it. Planning these tests is no small chore: Geodon's trials involved 15,000 patients at locations around the world. It took a year just to develop all of the necessary protocols. It took nearly three years to complete the testing.
Once more, Geodon appeared to deliver. Pfizer concluded that it was safe and effective-the two key ingredients of FDA approval. It was also an improvement over earlier products. "I had put on 100 pounds with other drugs," says Meghan Caughey, a 47-year-old patient who participated in Phase Three tests. "As soon as I started Geodon, I began to lose weight."
Pfizer believed it had a winner. On St. Patrick's Day in 1997, employees loaded a large Ryder truck with documents and two senior executives drove it to the FDA's headquarters in Maryland. For nearly a year, the FDA would study Geodon's test results and look for problems.
At Pfizer's R&D building in Groton, champagne bottles sat beside a fax machine on the day in February 1998 when the FDA had promised a ruling. Some 1,600 people at facilities around the world had worked on the drug for 15 years, and at long last they were going to win approval.
Or so they thought. But the letter from FDA announced that Geodon was not approved. "Everybody was blown away," says Zorn. This was the worst of all possible results, like a marathoner falling down a few yards shy of the finish line.
The FDA was concerned with something called the QTc interval, which, roughly speaking, is the amount of time it takes for a heart to contract and pump blood. The Phase Three trials had revealed that Geodon lengthens the QTc interval in patients, leading to a type of irregular heartbeat that can cause death. Pfizer had detected the problem, but didn't think the risk was significant. The FDA disagreed.
Pfizer was forced to confront the awful possibility that it had invested more than a decade of brainpower and hundreds of millions of dollars to develop a glorified rat poison. "It was frustrating to be turned down," says Swift, "because it felt like we had this drug that was really going to help a lot of people, but that we'd somehow stepped into a black hole."
At this point, Pfizer went back and studied the detailed patient narratives it had compiled in Phase Three. Company scientists determined that there was no evidence of increased heart problems among Geodon patients, even though the QTc interval had changed in many of them. So Pfizer decided to gamble: It would attempt to show that the QTc interval was no cause for alarm. This would require yet another test, and a complicated one at that. Pfizer located 150 healthy schizophrenics-a hard population to track down-and got them to agree to a massive study that would involve frequent blood tests and strict diets; the subjects would also have to live in a hospital. All of this was necessary, because Pfizer scientists were going to perform tests gauging changes in the heart rate that last mere milliseconds. "There was a great deal of debate about whether to proceed, because there were no guarantees," says Swift. "The company has a finite amount of resources, and the money invested in one project can't be used for another."
This last test took a year to complete-and the FDA took another year to examine the results. It then convened a non-binding advisory committee, which ruled heavily in Geodon's favor. Since the drug's patent had been established years earlier, every delay shortened the amount of time the company would have to recoup its investment in Geodon. Finally, on February 5, 2001, Geodon was approved, and Pfizer launched it the following month.
Since then, some 350,000 people have taken Geodon, mostly in the form of a little blue-and-white capsule. "We're seeing a dramatic difference in quality of life-schizophrenics can do things physically that they weren't able to do before," says Romano. That's good not only for patients, but for the rest of us, too. A study in the New England Journal of Medicine found that access to antipsychotic drugs like Geodon was closely linked to health-care costs; schizophrenics who don't take medication (or perhaps ignore their prescriptions to avoid side effects) are much more likely to require expensive hospital treatment.
Still, there's plenty left undone. About one-third of schizophrenics don't appear to be helped by Geodon, for reasons that aren't entirely clear. "It's also not a cure," says Jim Heym, a Pfizer vice president. With the advent of computer-aided drug design, plus full access to the human genome, researchers at Pfizer are hopeful that they'll continue to improve the treatment of schizophrenics. They also believe they're on the cusp of gains in fighting everything from Alzheimer's disease to cancer-but only if they're allowed to generate enough revenue for them to continue investing in the next generation of wonder drugs.
In the meantime, Geodon patients are just glad they can control their symptoms. Around the time of Geodon's release, Pfizer recorded a videotape of patients describing their problems-and how Geodon had improved their lives. Stevin Zorn pulled aside Harry Howard, the man who had built the first Geodon molecule, and who had moved on to other projects many years earlier. "I put the tape in the machine and pressed play," recalls Zorn. "The daughter of a woman named Brenda came on the screen. She had been asked what she would say to the creator of Geodon, if he were sitting right in front of you. She started to tear up, and exclaimed, 'I would grab him and hug him and they'd probably think I'm crazy but I do want to thank them from the bottom of my heart because I have my mom back.'" Harry Howard started to tear up, too.
COPYRIGHT 2003 National Review, Inc.
COPYRIGHT 2003 Gale Group