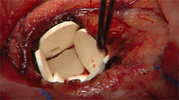
Gliadel
The GLIADEL® Wafer is a small wafer that contains the chemotherapeutic drug carmustine, or BCNU. After a specific brain tumor called a high-grade malignant glioma is surgically removed, up to 8 GLIADEL® Wafers are implanted in the cavity the tumor once occupied, slowly delivering BCNU directly to the tumor site. Find out more about GLIADEL® Wafer.
GLIADEL® is marketed by MGI® pharma more...
GLIADEL® Wafer is indicated in newly diagnosed high-grade malignant glioma patients as an adjunct to surgery and radiation. GLIADEL® Wafer is also indicated in recurrent glioblastoma multiforme patients as an adjunct to surgery.
The technology of the biocompatible and biodegradable polymer that constitutes the "body" of the wafer was developed by the MIT (Massachussets Institute of Thecnology).
GLIADEL® Wafer and survival
A recent clinical study was conducted in 240 men and women undergoing initial surgery for a newly diagnosed high-grade malignant glioma. Each patient was randomly assigned to receive either surgery with implantation of GLIADEL® Wafer followed by radiation therapy, or surgery with implantation of placebo wafers (wafers without any BCNU) followed by radiation therapy.
The results in this study showed that survival was significantly prolonged in the patients who received GLIADEL® Wafer compared to those who received the placebo wafers: median survival—the length of time half of the patients in the study survived—increased to 13.9 months from 11.6 months.
Patients undergoing craniotomy and implantation of GLIADEL® Wafer for malignant glioma should be monitored closely for complications of craniotomy. During a randomized trial of GLIADEL® Wafer vs placebo implanted during initial resection, five categories of adverse events occurred that are possibly treatment-related: seizure (33.3% vs 37.5%); brain edema (22.5% vs 19.2%); healing abnormalities (15.8% vs 11.7%) including cerebrospinal fluid (CSF) leaks (5.0% vs 0.8%); intracranial hypertension (9.0% vs 2.0%); and intracranial infection (5.0% vs 6.0%). During a randomized trial of GLIADEL® Wafer vs placebo for recurrent disease, five categories of adverse events occurred that are possibly treatment-related: post-operative seizure (19.0% vs 19.0%); healing abnormalities (14.0% vs 5.0%); intracranial hypertension (4.0% vs 6.0%); brain edema (4.0% vs 1.0%); and intracranial infection (4.0% vs 1.0%).
Cases of intracerebral mass effect unresponsive to corticosteroids have been described in patients treated with GLIADEL® Wafer, including one case leading to brain herniation. GLIADEL® Wafer contains carmustine and should not be given to patients who are allergic to carmustine. There are no studies assessing the reproductive toxicity of GLIADEL® Wafer. Carmustine can also cause fetal harm when administered to a pregnant woman.
- PI
- FAQ's for patients
- Clinical trials and noteworthy treatments for brain tumors
Read more at Wikipedia.org