Guilford Pharmaceuticals Inc., (NASDAQ: GLFD) announced today that it
has filed a submission in Europe seeking an expanded indication for
its innovative cancer therapy, GLIADEL(R) Wafer, for use during
initial surgery for malignant glioma. Currently, GLIADEL(R) Wafer,
is approved in 24 countries, including the United States, for use
during recurrent surgery for glioblastoma multiforme (GBM), a rapidly
fatal form of malignant brain cancer, and in Canada for use in both
primary and recurrent surgery for GBM.
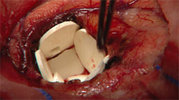
GLIADEL(R) Wafer is a biodegradable wafer, implanted at the time of
surgery, that delivers chemotherapy directly to the tumor site,
minimizing drug exposure to other areas of the body. GLIADEL(R)
Wafer complements other standard therapies for brain cancer, such as
surgery, radiation and traditional intravenous chemotherapy.
"Our European submission is an important commercial milestone for the
Company," commented Craig R. Smith, M.D., President and Chief
Executive Officer of Guilford. "In Europe, the surgical treatment of
malignant glioma is often restricted to initial surgery; with
recurrent surgeries occurring much less frequently than in the United
States. Obtaining marketing authorization for GLIADEL(R) Wafer for
use during initial surgery in Europe creates a much broader market
opportunity for the product than presently exists, and second,
provides improved treatment options for patients with this
devastating disease."
The European regulatory submission is based on favorable results from a
randomized, double-blind, placebo-controlled Phase III clinical
trial, which demonstrated a 30 percent improvement in survival rates
in patients who received GLIADEL(R) Wafer therapy at the time of
their initial diagnosis and surgery for malignant glioma.
In the study, at one year, 59 percent of patients given GLIADEL(R)
Wafer were still alive compared to 48 percent of those given placebo.
A secondary endpoint in the study - improvement in neurological
symptoms - also demonstrated a statistically significant benefit in
the GLIADEL(R) Wafer treatment group.
GLIADEL(R) Wafers have generally been well tolerated. However,
GLIADEL(R) Wafer contains carmustine and should not be given to
patients who are allergic to carmustine. Carmustine can also cause
fetal harm when administered to a pregnant woman. Patients
undergoing surgery for malignant brain cancer and implantation of
GLIADEL(R) Wafer should be monitored closely for the following known
complications of surgery including: seizures, intracranial
infections, abnormal wound healing, and brain edema. Each of these
complications has been associated with GLIADEL(R) Wafer treatment.
About GLIADEL(R) Wafer
GLIADEL(R) Wafer is a small, white dime-sized wafer made of a
biodegradable polymer that contains the cancer chemotherapeutic drug,
carmustine or BCNU. Up to eight GLIADEL(R) Wafers can be implanted
in the cavity created when a brain tumor is surgically removed.
There, the wafers slowly dissolve over a period of 2 to 3 weeks,
delivering chemotherapy directly to the tumor site in high
concentrations, while minimizing drug exposure to other areas of the
body.
Guilford Pharmaceuticals Inc. is a global, fully-integrated
pharmaceutical company targeting the neurological, surgical and
critical care markets. The Company's mission is to develop
proprietary biopolymer-based therapeutics for surgeons and novel
pharmaceutical products for the diagnosis and treatment of
neurological disorders.
For more information about GLIADEL(R) Wafer, visit www.gliadel.com.
For more information about Guilford Pharmaceuticals, visit
www.guilfordpharm.com
This press release contains forward-looking statements that involve
risks and uncertainties, including those described in the section
entitled "Risk Factors" contained in the Company's Registration
Statement on Form S-3 filed with the SEC on June 21, 2001 (File No.
333-63576). Among other things, there can be no assurance that the
European regulatory agencies will approve GLIADEL(R) Wafer for
initial therapy in patients newly diagnosed with malignant glioma, or
that if approved GLIADEL(R) Wafer will achieve market acceptance for
the expanded indication.