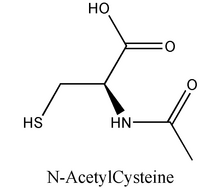
Study objective: To study the effect of the antioxidant N-acetylcysteine (NAC) in the development of elastase-induced emphysema in rats.
Materials and methods: Wistar rats (n = 72) were orotracheally instilled with 75 IU elastase or saline solution. Eighteen rats from each group received the antioxidant NAC from 2 days before induction of the lesion until they were killed 2, 8, and 28 days after instillation. The effects of treatment were assessed by measuring collagen content for the left lung, a histopathology evaluation (ie, mean alveolar internal surface area (AIA) and mean linear intercept measurement), and lung function.
Results: Twenty-eight days after elastase instillation, rats treated with NAC showed significant attenuation of the lesion in comparison with rats treated only with elastase, including the following: normalization of mean ([+ or -] SEM) collagen content (1.23 [+ or -] 0.09 vs 1.51 [+ or -] 0.10 mg per left lung, respectively; p < 0.05); partial inhibition of mean AIA (14,860 [+ or -] 1,135 vs 19,622 [+ or -] 1,294 [micro][m.sup.2], respectively; p < 0.05) and mean linear intercept (108.8 [+ or -] 3.7 vs 123.0 [+ or -] 4.2 [micro]m, respectively; p < 0.05); and increases and improvement in expiratory flows (27.8 [+ or -] 1.2 vs 23.4 [+ or -] 1.3 mL/s, respectively; p < 0.05). NAC was not able to avoid the compliance increase in the elastase-plus-NAC group.
Conclusion: Consistent with the results of anatomic, pathologic, and functional studies, NAC is able to attenuate the lesions induced by elastase in rats, which is in accordance with previous data supporting the idea that oxidant injury could contribute to the development of elastase-induced emphysema.
Key words: elastase; emphysema: lung function; N-acetylcysteine: rats
Abbreviations: AIA = alveolar internal surface area: CL = lung compliance; F75 = expiratory flow at 75% of FVC; HYP = hydroxyproline; IC = inspiratory capacity; Lm = mean linear intersection: Lmc = computerized mean linear intersection NAC = N-acetycyteine; PMN = polymorphonuclear cell
*********
Intratracheal elastase administration induces a lesion that resembles human panacinar emphysema. (1) In hamsters, elastase initially provokes a severe decrease in the elastin content of the lung that is gradually recovered while the emphysema worsens. (2) There also appears to be an increase in collagen synthesis during the development of emphysema. (2) These changes in connective tissue alter elastase administration, observed not only biochemically but also by electron microscopy, (3) reflect the repair reaction of the lung after elastase-induced injury.
Functionally, the lesion produces an increase of inspiratory capacity (IC) and lung distensibility, and a decrease of expiratory flows. Morphologically, elastase provokes a disruption of the alveolar walls that leads to the enlargement of the airspace regularly distributed throughout all the parenchyma, which is reflected in an increase of the mean linear intercept. (4) Furthermore, intratracheal elastase induces an early inflammatory response with neutrophils and macrophages, (2) which is still present 1 month later. (3) These cells could be the source of proteases and oxidants that can contribute to the destruction of lung connective tissue as well as of inflammatory mediators that exacerbate elastase-induced emphysema. (5)
N-acetylcysteine (NAC) is a precursor of glutathione molecules and has oxygen radical-scavenging properties. (6,7) The effectiveness of NAC administration in animal models of lung fibrosis as well as in patients with idiopathic pulmonary fibrosis has been reported. In this respect, it has been demonstrated (8-10) that NAC is able to attenuate cellular infiltration and collagen deposition in a model of bleomycin-induced lung fibrosis. When administered together with steroids, it improves the lung function index in patients with idiopathic pulmonary fibrosis. (11) It also has been reported that NAC has an anti-inflammatory role because of its capacity to regulate the production of some inflammatory mediators in fibrosis induced in vitro (12) and in vivo. (10) To our knowledge, there are no studies about the effect of NAC administration on the evolution of induced pulmonary emphysema. The aim of this study was to investigate whether oral administration of antioxidant/anti-inflammatory NAC has any effect on the course of development over time of elastase-induced emphysema in rats in terms of repair, lung function, and morphometry.
MATERIALS AND METHODS
Materials and Subjects
The Animal Research Committee of the center approved all animal experimentation. Male Wistar rats (weight range, 180 to 200 g) were classified into the following four groups: a control group (n = 18) orotracheally instilled with saline solution; an elastase group (n = 18) orotracheally instilled with 75 IU porcine pancreatic elastase (Boche Diagnostics; Basel, Switzerland); a control group treated with NAC (n = 18); and an elastase group treated with NAC (n = 18). After instillation, the rats were returned to their cages. The rats that were to receive NAC (Laboratorios Zambon SA; Barcelona, Spain) were given 200 mg NAC per rat per day mixed with powdered food divided into two doses (at 9:00 AM and at 6:00 PM) beginning 2 days before the instillation until the study day. Food was provided ad libitum, checking that there was as little food as possible remaining between dosages in the groups receiving NAC. The dosage, which was equivalent to the human dosage in terms of body surface, was the same that proved to be effective ameliorating cigarette smoke induced lung lesions in rats when used previously. (13) Rats were killed 2, 8, and 28 days after instillation.
Lung Function Tests
The functional study was performed in a breathing assembly for small animals, as described elsewhere. (4) The rats were anesthetized with sodium pentobarbital (60 mg/kg intraperitoneally), tracheotomized, put into a plethysmograph, and fitted to a cannula that allowed communication with the breathing assembly. As soon as the rat was connected to the breathing equipment, it was paralyzed with 0.2 mg pancuronium bromide and then artificially ventilated. Changes in IC, lung compliance (CL), and expiratory flow at 75% of FVC (F75) were assessed.
Morphometry
After lung function study, the chest was opened. The left lung was weighed and immediately frozen for the biochemical determination of collagen (see below). The right lung was fixed by filling it with 10% formalin to an airway pressure of 25 cm [H.sub.2]O fur 24 h. Five-micrometer sections were stained with hematoxylin-eosin. The mean linear intersection (Lm) was measured as previously described. (4) Using computer-assisted imaging analysis, the mean of the horizontal and vertical alveoli lengths (ie, the mean computerized linear intersection [Lmc]), equivalent to the Lm, as well as the mean alveolar internal surface area (AIA) were calculated in each field. Images were visualized by a video camera (Leica DC 100; Leica Microsystems; Cambridge, UK) with a resolution of 782 x 582 pixels adapted to a microscope (model BX40; Olympus; Tokyo, Japan). Routine imaging analysis software (Qwin; Leica Microsystems) adapted for the purpose of this study was applied for AIA and Line determinations. Fields (12 to 18 per rat) were quantified under a x4 objective and a x0.5 reducing video camera adapter. For each animal, the AIA and Lmc were averaged from all the fields measured.
Collagen Analysis
Collagen quantification was performed by measuring hydroxyproline (HYP). Briefly, the left lung was homogenized, and, after acid hydrolysis, chloramine T (Merck; Darmstadt, Germany) was added to the hydrolyzate to allow for oxidation. After the addition of the Ehrlich reagent (Sigma; St. Louis, MO) and reaction development, absorption of each sample was read at 560 nm. (14) The results are expressed in milligrams of HYP per left lung.
Statistical Analysis
All data are expressed as the mean [+ or -] SEM. Comparisons were made with analysis of variance. Multiple range tests (least significance differences method) were used for the analysis of differences among means. Pearson correlations also were calculated.
RESULTS
Collagen Content
The sequential results of collagen changes over time are represented in Figure 1. Collagen measured by HYP was significantly higher in elastase-treated rats at 8 days, with the difference increasing with respect to the control group alter 28 days. Treatment with NAC significantly hindered the increase of HYP from the first days 'after instillation, which was significantly lower than that in the elastase group on the 28th day (1.23 [+ or -] 0.09 vs 1.51 [+ or -] 0.1 mg, respectively) and was not significantly different from that of the control group at any point in time.
[FIGURE 1 OMITTED]
Morphometry
In terms of airspace enlargement, the lesion was still not evident on or before the 8th day. For this reason, we have focused on Lm and area values after 28 days (Table 1). Rats treated with elastase presented significantly increased AIA compared with that of the control group. As shown in Figure 2, the elastase-plus-NAC group showed significantly lower percentages of Line and AIA than the elastase group.
[FIGURE 2 OMITTED]
Figure 3 shows histopathologic images of lung sections from the control, elastase, and elastase-plus-NAC groups obtained 28 days after induction of the lesion. The Lm followed the same tendency and correlated highly with Lmc (r = 0.96; p < 0.0,5).
[FIGURE 3 OMITTED]
The correlation between AIA and HYP also was calculated. As can be seen in Figure 4, 28 (lays after elastase administration, a significant positive correlation was found between AIA increase and HYP level in the elastase and control groups (r = 0.46; p < 0.0,5), but not in the elastase-plus-NAC group.
[FIGURE 4 OMITTED]
Functional Study
The course of function deterioration over time in the control, control-plus-NAC, elastase, and elastase-plus-NAC groups for IC, CL, and F75 percentages are represented in Figures 4 and 5. IC was significantly lower in the first days after elastase administration, but was not different from the saline control group after 28 days (Fig 5, left, A). CL (Fig 5, right, B) was not different from the control group in the first days after elastase administration, but was significantly higher in both elastase groups on the 28th day. As can be seen in Figure 6, F75, expressed as the percentage of decline with respect to the control group, was significantly decreased in the elastase and elastase-plus-NAC groups after 2, 8, and 28 days. However, on the 28th day the decline was significantly lower in the elastase-plus-NAC group compared with the elastase group. Correlations between functional and morphometric parameters after 28 days were calculated for the elastase and elastase-plus-NAC groups. As shown in Table 2, AIA correlated positively with CL and negatively with F75 in both elastase groups. In the elastase-plus-NAC group, the correlation with CL was higher and that with F75 was lower than those obtained in the elastase group, in absolute values. Line followed the same tendency.
[FIGURES 5-6 OMITTED]
DISCUSSION
Treatment with oral NAC partially attenuates lung emphysema induced by elastase in rats. Improvement is observed in the amelioration of airspace enlargements, the partial recovery of expiratory flows, and the normalization of lung collagen content.
We found that after elastase administration there was an evident increase of collagen from the 8th day. The higher collagen content in elastase-treated animals was initially found by Kuhn et al (2) in hamsters, and later this event was described with detail using scanning electron microscopy in rats. (3) The alveolar structures of the lung seem to initiate repair responses after structural injury. In this way, elastin and collagen gene expression is up-regulated after elastase administration. (15) There is some evidence suggesting that inadequate repair contributes to the worsening of emphysema. For example, starvation, which generally inhibits anabolic responses, can exacerbate the development of elastase-induced emphysema in animals. (16) In addition, the inhibition of elastin synthesis by [beta]-aminopropionitrile, a lathyrogen that inhibits elastin maturation, worsens emphysema in elastase-exposed animals. (17) Our data demonstrate that the repair reaction started after elastase administration does not, however, efficiently restore tissue integrity. Morphometric determinations after 28 days showed that elastase provoked an enlargement of alveolar spaces reflected in an increase of AIA of 280% with respect to the control group, which is compatible with the increase in Lmc. Moreover, we found that the higher amount of HYP in the elastase group correlated positively with airspace enlargement, quantified in terms of AIA (r = 0.46) [Fig 3]. In accordance with these results, Vlahovic et al (18) found a positive correlation between Lm and alveolar interstitial thickness in patients with emphysema. Thus, there seems to be a relationship between the degree of parenchyma destruction and the synthesis of collagen for reparation. Regarding lung function, after 2 and 8 days there was a decrease of IC, CL, and F75, probably reflecting lung edema produced after elastase instillation. Thereafter, the lesions developed, and, on the 28th day, there was an increase of CL and a fall in expiratory flows without changes in IC, which was similar to the results obtained in previous studies. (4,19)
Interestingly, the daily administration of NAC for 28 days after elastase instillation significantly reduced lung collagen content compared to the elastase group. Morphometrically, the emphysema was less severe, with both AIA and Lmc being significantly lower than that in the elastase group (reduction of 25% for AIA and 12% for Lmc compared to elastase group). Functionally, expiratory flow was significantly improved after NAC treatment, although values still remained lower than those in the control group. However, CL was not influenced by NAC and was equally higher in both elastase groups, suggesting that NAC does not interfere in the elastolytic process. Nevertheless, the correlation between CL and AIA (Table 2) not only persisted in the elastase-plus-NAC group but was even higher than that for the elastase group. This could mean that in the elastase-plus-NAC group the increased distensibility is mainly explained by alveolar enlargement. In contrast, in the elastase group several other factors could be involved that may interfere in the relationship between the increase in AIA and distensibility. Taking into account the lack of change in CL, the improvement of expiratory flow in the elastase-plus-NAC group only could be explained by an increase in airway conductance in rats treated with NAC. In a similar study, retinoic acid was administered daily for 2 weeks, 25 days after elastase instillation. However, despite notable lung morphology results, (20) retinoic acid was not able to reverse either increased CL or expiratory flows (21) and anatomic-functional correlations were not stated.
Is difficult to believe that NAC prevents the direct enzymatic effect of elastase. It must be acting at a different level. Previous studies have demonstrated that 2 h after elastase administration its activity is markedly reduced (22) and is cleared from the lung within 24 h. (23) However, the development of emphysema is gradual over a span of at least 2 months, (22) suggesting that endogenous mechanisms must be involved.
It has been reported that during the first 48 to 72 h after elastase administration there is a moderate acute inflammatory response with neutrophils and macrophages, (2) and that 4 weeks later there is still a markedly elevated total cell count. (3) Activated inflammatory cells can release oxygen radicals and proteases that can directly damage components of the lung matrix. Moreover, it has been demonstrated (24) that several proteases induce a direct rise in reactive oxygen species in bronchial epithelial cells and fibroblasts. On the other hand, Left et al (25) described a decrease in neutrophil influx into lung lavage fluid after NAC administration in all animal model treated with interleukin-1[alpha].
In addition, genes for many inflammatory mediators, some of them involved in the recruitment of inflammatory cells and, therefore, in the development of emphysema, (5) are regulated by transcription factors such as nuclear factor [kappa]B. Nuclear factor [kappa]B activation can be induced by oxidants, (26) and NAC previously has been demonstrated to be efficient in decreasing this activation in vivo. (27) In the elastase-induced emphysema model, NAC could be acting at this level by hindering the overexpression of genes for inflammatory cytokines.
Thus, lesions induced in rats by elastase instillation seem to be not only the consequence of the initial enzymatic damage but, more probably, the result of several secondary stimuli that lead to the worsening of the disease over time. Lucey et al (5) recently stated that tumor necrosis factor-[alpha] and interleukin-1[beta] account for about 80% of the emphysema that develops after elastase treatment, pointing out that these proinflammatory mediators (polymorphonuclear cell [PMN] chemoattractants) are taking pact in the development of the lesion. But actually, there is no evidence showing that the elastase-induced emphysema is accompanied by an increase in the number of PMN cells. According to these data, Cantor et al (28) found that 1 week after the induction of the lesion with elastase the number of PMNs was normal. In our experiment, it is not possible to know whether NAC treatment completely abolishes the oxidative stress occurring after elastase, but we think that oxidants can explain at least part of the emphysematous lesion, taking into account the possible anti-inflammatory effect of NAC throughout the diminution of PMN influx into the lung.
The effectiveness of NAC in attenuating bleomycin-induced lung fibrosis is unquestionable, (8-10) although the mechanism by which it limits fibrosis is also unclear. Bleomycin damage consists of the following two phases: an early inflammatory phase, characterized by cellular infiltration; and a late fibrotic phase. Reactive oxygen species and proteases generated from infiltrated cells are considered to injure the lung tissue, and excessive fibrosis occurs as a reparative process. These two phases also seem to be present in emphysema development, the second chronic severe reparative phase establishing the different pathophysiologies of both diseases.
In patients with COPD, the oxidant burden is also increased. Cigarette smoke, the major etiologic factor in COPD, is a rich source of oxidant molecules. Oxidative stress is also a critical feature in the pathogenesis of COPD since it results in the inactivation of antiproteinases, airspace epithelial injury, mucus hypersecretion, increased influx of neutrophils into the lungs, transcription factor activation, and gene expression of proinflammatory mediators. Antioxidants, therefore, should not only protect against the direct injurious effects of oxidants, but may also fundamentally alter the inflammatory events that have a central role in the pathogenesis of COPD. (29)
Fibrosis, emphysema, and COPD seem to have the oxidant/antioxidant imbalance in common. The site and specific characteristics of the inflammatory responses may differ in each of these diseases.
In conclusion, oxidant/antioxidant imbalance seems to contribute to the development of elastase-induced emphysema, and treatment with antioxidant NAC could attenuate or slow down the process.
REFERENCES
(1) Snider GL, Lucey EC, Stone PJ. Animal models of emphysema. Am Rev Respir Dis 1985; 133:149-169
(2) Kuhn C, Yu S-Y. Chraplyvy MS. et al. The induction of emphysema with elastase: changes in connective tissue. Lab Invest 1976; 34:372-380
(3) Finlay GA, O'Donnell MD, O'Connor CM. et al. Elastin and collagen remodeling in emphysema: a scanning electron microscopy study. Am J Pathol 1996; 149:1405-1415
(4) Rubio ML. Sanchez-Cifuentes MV, Peces-Barba G, et al. Intrapulmonary gas mixing in panacinar and centriacinar induced emphysema in rats. Am J Respir Crit Care Med 1998; 157:237-245
(5) Lucey EC, Keane J, Kuang P-P, et al. Severity of elastase-induced induced emphysema is decreased in tumor necrosis factor-[alpha] and interleukin-1[beta] receptor-deficient mice. Lab Invest 2002; 82:79-85
(6) Aruoma OI, Halliwell B, Hoey BM, et al. The antioxidant action of N-acetylcysteine: its reaction with hydrogen peroxide, hydroxyl radical, superoxide and hypochlorous acid. Free Radio Biol Med 1989; 6:593-597
(7) Gillisen A, Nowak D. Characterization of N-acetylcysteine and ambroxol in anti-oxidant therapy. Respir Med 1998; 92:609-623
(8) Cortijo J, Cerda-Nicolas M, Serrano A. et al. Attenuation by oral N-acetylcysteine of bleomycin-induced lung injury in rats. Eur Respir J 2001; 17:1228-1235
(9) Shahzeidi S, Sarnstrand B, Jeffery PK, et al. Oral N-acetylcysteine reduces bleomycin-induced collagen deposition in the lungs of mice. Eur Respir J 1991; 4:845-852
(10) Hagiwara S-I, Ishii Y, Kitamura S. Aerosolized administration of N-acetylcysteine attenuates lung fibrosis induced by bleomycin in mice. Am J Respir Crit Care Med 2000; 162:225-231
(11) Behr J, Maier K, Degenkolb B, et al. Antioxidative and clinical effects of high-dose N-acetylcysteine in fibrosing alveolitis: adjunctive therapy to maintenance immunosupression. Am J Respir Crit Care Med 1997; 156:1897-1901
(12) Gon Y, Hashimoto S, Nakayama T, et al. N-acetyl-L-cysteine inhibits bleomycin induced interleukin-8 secretion by bronchial epithelial cells. Respirology 2000; 5:309-313
(13) Rubio ML, Sanchez-Cifuentes MV, Ortega M, et al. N-acetycysteine prevents cigarette smoke induced small airways alterations in rats. Eur Respir J 2000; 15:505-511
(14) Reddy KG, Enwemeka CS. A simplified method for the analysis of hydroxyproline in biological tissues. Clin Biochem 1996; 29:225-229
(15) Lucey EC, Goldstein RH. Stone PJ, et al. Remodeling of alveolar walls after elastase treatment of hamsters: results of elastin and collagen mRNA in situ hybridization. Am J Respir Crit Care Med 1998; 158:555-564
(16) Sahebjami H, Wirman JA. Emphysema-like changes in the lungs of starved rats. Am Rev Respir Dis 1981; 124:619-624
(17) Kuhn C, Starcher BC. The effect of lathyrogens on the evolution of elastase-induced emphysema. Am Rev Respir Dis 1980; 122:453-460
(18) Vlahovic G, Russell ML, Mercer RR, et al. Cellular and connective tissue changes in alveolar septal walls in emphysema. Am J Respir Crit Care Med 1999; 160:2086-2092
(19) Gonzalez Mangado N, Peces-Barba G, Cabanillas JJ, et al. Effect on single-breath washout and lung function of elastase-induced emphysema in rats. Am Rev Respir Dis 1993; 148:735-743
(20) Massaro GD, Massaro D. Retinoic acid treatment abrogates elastase-induced pulmonary emphysema in rats. Nat Med 1997; 3:675-677
(21) Tepper J, Pfeiffer J, Aldrich M, et al. Can retinoic acid ameliorate the physiologic and morphologic effects of elastase instillation in the rat? Chest 2000; 117:242S-244S
(22) Kaplan PD, Kuhn C, Pierre JA. The induction of emphysema with elastase: I. The evolution of the lesion and the influence of serum. J Lab Clin Med 1973; 82:349-356
(23) Stone PJ, Lucey EC, Calore JD, et al. Defenses of the hamster lung against human neutrophil and porcine pancreatic elastase, Respiration 1988; 54:1-15
(24) Aoshiba K, Yasuda K, Yasui S, et al. Serine proteases increase oxidative stress in lung cells. Am J Physiol Lung Cell Mol Physiol 2001; 281:L556-L564
(25) Leff JA, Wilke CP, Hybertson BM, et al. Postinsult treatment with N-acetyl-L-cysteine decreases IL-1 induced neutrophil influx and lung leak in rats. Am J Physiol 1993; 265:L501-L506
(26) Rahman I, MacNee W. Role of transcription factors in inflammatory lung diseases. Thorax 1998; 53:601-612
(27) Rubio ML, Martin-Mosquero MC, Ortega M, et al. N-acetylcysteine suppresses cigarette smoke-induced nuclear factor-[kappa]Bba activation [abstract]. Eur Respir J 2000; 16:256s
(28) Cantor JO, Cerreta JM, Armand G, et al. Aerosolized hyaluronic acid decreases alveolar injury induced by human neutrophil elastase. Proc Soc Exp Biol Med 1998; 217:471-475
(29) MacNee W, Rahman I. Is oxidative stress central to the pathogenesis of chronic obstructive pulmonary disease? Trends Mol Med 2001; 7:55-62
* From the Laboratorio Neumologia Experimental, Servicio de Neumologia, Fundacion Jimenez Diaz, Universidad Autonoma, Madrid, Spain.
Supported in part by Zambon SA and Red Respira (grant RTIC C03/11,FIS,ISCIII).
Manuscript received March 25, 2003; revision accepted September 1, 2003.
Reproduction of this article is prohibited without written permission from the American College of Chest Physicians (e-mail: permissions@chestnet.org).
Correspondence to: Nicolas Gonzalez Mangado, MD, PhD, Servicio de Neumologia, Fundacion Jimenez Diaz Avdal Reyes Catolicos. 2 28040-Madrid, Spain; e-mail: ngonzalez@fjd.es
COPYRIGHT 2004 American College of Chest Physicians
COPYRIGHT 2004 Gale Group