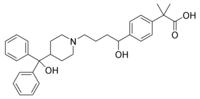
WOODCLIFF LAKE, N.J. -- Barr Pharmaceuticals will be allowing Teva to launch a generic Allegra product within its 180-day exclusivity period in exchange for a percent of the profits, the two companies announced Sept. 6. However, neither company is expected to field a generic version of Sanofi-Aventis' prescription antihistamine until early 2006, following the resolution of a patent dispute between Sanofi-Aventis and the generic drug suppliers.
"The courts have yet to resolve the pending patent litigation concerning our generic products," stated Barr chairman and chief executive officer Bruce Downey. "The agreement with Teva enables us to maximize the opportunity, while sharing the risk of the ongoing litigation."
The week following the Barr/Teva agreement, Sanofi-Aventis entered a distribution and supply agreement with Prasco Laboratories to begin providing an authorized generic version of Allegra products as soon as Sept. 19. "Sanofi-Aventis continues to believe that the generic product of Barr Pharmaceuticals and Teva Pharmaceuticals infringes its intellectual rights.
... Sanofi-Aventis will continue to vigorously defend its rights," the company stated in a press release.
In June 2004, Barr and Teva were granted summary judgment of non-infringement with respect to three patents and, in April 2005, were granted summary judgment of invalidity on an additional patent in the case. Several patents remain in litigation.
Although no trial date has been set, the companies expect that a trial will occur sometime in early 2006. Barr was granted final approval by the Food and Drug Administration for its generic Allegra earlier this month.
Allegra tablets had annual sales of approximately $1.4 billion, based on IMS data for the 12 months ended in June 2005, the companies reported.
COPYRIGHT 2005 Reproduced with permission of the copyright holder. Further reproduction or distribution is prohibited without permission.
COPYRIGHT 2005 Gale Group