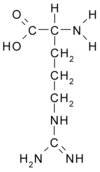
Introduction
L-Arginine is a semi-essential amino acid involved in numerous areas of human physiology, including production of nitric oxide (NO)--a key messenger molecule involved in vascular regulation, immune activity, and endocrine function. Arginine is also involved in protein production, wound healing, erectile function, and fertility. Arginine is not considered essential because humans can synthesize it de novo from glutamine, glutamate, and proline. However, dietary intake remains the primary determinant of plasma arginine levels, since the rate of arginine biosynthesis does not compensate for depletion or inadequate supply. (1,2)
Arginine is the most abundant nitrogen carrier in humans, containing four nitrogen atoms per molecule. Arginine is not a major inter-organ nitrogen shuttle; instead, it plays an important role in nitrogen metabolism and ammonia detoxification as an intermediate in the urea cycle. (3)
Biochemistry
Arginine is synthesized in mammals from glutamine via pyrroline 5-carboxylate (P5C) synthase and proline oxidase in a multi-step metabolic conversion. (4) In adults, most endogenous arginine is produced from citrulline, a by-product of glutamine metabolism in the gut and liver. Citrulline is released into the circulation and taken up primarily by the kidney for conversion into arginine. (5)
Supplemental arginine is readily absorbed. (6) About 50-percent of ingested arginine is rapidly converted in the body to ornithine, primarily by the enzyme arginase. (7) Because of this fast turnover, sustained-release preparations are being investigated as a way to maintain a steadier blood level over time. Ornithine, in turn, can be metabolized to glutamate and proline, or through the enzyme ornithine decarboxylase into the polyamine pathway for degradation into compounds such as putrescine and other polyamines.
In addition, arginine is a precursor for the synthesis of nitric oxide, proteins, urea, creatine, vasopressin, and agmatine. (8) Arginine that is not metabolized by arginase to ornithine is processed by one of four other enzymes: nitric oxide synthase (to become nitric oxide); arginine:glycine amidinotransferase (to become creatine); arginine decarboxylase (to become agmatine); or arginyl-tRNA synthetase (to become arginyl-tRNA, a precursor to protein synthesis). Arginine is also an allosteric activator of N-acetylglutamate synthase, which synthesizes N-acetylglutamate from glutamate and acetyl-CoA. (9)
Mechanisms of Action
Arginine is the biological precursor of nitric oxide (NO), an endogenous gaseous messenger molecule involved in a variety of endothelium-dependent physiological effects in the cardiovascular system. (10) Much of arginine's influence on the cardiovascular system is due to endothelial NO synthesis, which results in vascular smooth muscle relaxation and subsequent vasodilation, as well as inhibition of monocyte adhesiveness, platelet aggregation, and smooth muscle proliferation. A great deal of research has explored the biological roles and properties of nitric oxide, (11,12) which is also of critical importance in maintenance of normal blood pressure, (13) myocardial function, (14) inflammatory response, (15) apoptosis, (16) and protection against oxidative damage. (17)
Arginine is a potent immunomodulator. Supplemental arginine appears to up-regulate immune function and reduce the incidence of postoperative infection. Significant decreases in cell adhesion molecules and pro-inflammatory cytokine levels have also been observed. Arginine supplementation (30 g/day for three days) has been shown to significantly enhance natural killer (NK) cell activity, lymphokine-activated killer cell cytotoxicity, and lymphocyte mitogenic reactivity in patients with locally advanced breast cancer. (18,19)
Arginine has significant effects on endocrine function--particularly adrenal and pituitary secretion--in humans and animals. Arginine administration can stimulate the release of catecholamines, (20) insulin and glucagon, (21) prolactin, (22) and growth hormone (GH); (23,24) however, little is known about the specific mechanism(s) by which arginine exerts these effects.
Clinical Indications
Cardiovascular Conditions
Arginine's effects on cardiovascular function are due to arginine-induced endothelial NO production. Endothelial nitric oxide synthase (eNOS) catalyzes this reaction, which produces NO and ornithine. Nitric oxide diffuses into the underlying smooth muscle and stimulates guanylyl cyclase, producing guanosine-3',5'-cyclic monophosphate (cGMP), which in turn causes muscle relaxation and vasodilation. Arginine supplementation has been shown to increase flow-mediated brachial artery dilation in normal individuals as well as those with hyperlipidemia and hypertension. (25,26) Nitric oxide is also responsible for creating an environment in the endothelium that is anti-atherogenic. Adequate NO production inhibits processes at the core of the atherosclerotic lesion, including platelet aggregation, monocyte adhesion and migration, smooth muscle proliferation, and vasoconstriction.
Asymmetrical dimethylarginine (ADMA) competes with arginine for binding with eNOS, subsequently down-regulating activity of this vital enzyme. Increased plasma ADMA has been shown to be an independent risk factor for cardiovascular disease because of its inhibitory activity on eNOS. Oral arginine supplementation overrides the inhibitory effect of ADMA on eNOS, and improves vascular function in those with high ADMA levels. (27-29)
Angina Pectoris
Arginine supplementation has been effective in angina treatment in some, but not all, clinical trials. In 36 patients with chronic, stable angina given 6 g arginine daily for two weeks, significant improvement was noted in flow-mediated vasodilation, exercise time, and quality of life, compared to placebo. No improvement was seen in ischemia markers on ECG or in time-to-onset of angina. (30)
In a small, uncontrolled trial, seven of 10 people with intractable angina improved dramatically after taking 9 g arginine daily for three months. (31) A double-blind trial in 22 patients with stable angina and healed myocardial infarction showed oral supplementation with 6 g arginine daily for three days increased exercise capacity. (32) However, in men with stable angina, oral supplementation with arginine (15 g/day) for two weeks was not associated with improvement in endothelium-dependent vasodilation, oxidative stress, or exercise performance. (33) In patients with coronary artery disease, oral supplementation of arginine (6 g/day for three days) did not affect exercise-induced changes in QT interval duration, QT dispersion, or the magnitude of ST-segment depression; (34) however, it did significantly increase exercise tolerance. The therapeutic effect of arginine in patients with microvascular angina is considered to be the result of improved endothelium-dependent coronary vasodilation. (35)
Congestive Heart Failure
Six weeks of oral arginine supplementation (5.6-12.6 g/d) significantly improved blood flow, arterial compliance, and functional status in patients with congestive heart failure (CHF), compared to placebo, in a randomized, double-blind trial. (36) Another double-blind trial found arginine supplementation (5 g three times daily) improved renal function in people with CHF. (37) After a one-week oral dosing with 6 g arginine daily in 30 males with stable CHF, significant improvements were seen in exercise duration, anaerobic threshold, and V[O.sub.2]. (38) African Americans are at significantly greater risk for development of CHF than Caucasians. However, the improvement in endothelial function seen with arginine dosing may be more pronounced in African Americans compared to Caucasians, as was seen in a study of 52 CHF patients treated with an intra-coronary infusion of arginine. (39)
Hypertension
Administration of arginine prevented hypertension in salt-sensitive rats, but not in spontaneously hypertensive rats. (40) If arginine was provided early, hypertension and renal failure could be prevented. In healthy human subjects, intravenous (IV) administration of arginine had vasodilatory and antihypertensive effects. (41) In a small, controlled trial, hypertensive patients refractory to enalapril and hydrochlorothiazide responded favorably to the addition of oral arginine (2 g three times daily). (42) Small, preliminary trials have found oral (43) and IV (44) arginine significantly lowers blood pressure in healthy volunteers.
IV infusion of arginine (15 mg/kg body weight/min for 35 min) improved pulmonary vascular resistance index and cardiac output in infants with pulmonary hypertension. (45)
Intermittent Claudication
Intravenous arginine injections significantly improved symptoms of intermittent claudication in a double-blind trial. Eight grams of arginine, infused twice daily for three weeks, improved pain-free walking distance by 230 [+ or -] 63 percent and the absolute walking distance by 155 [+ or -] 48 percent (each p < 0.05) compared to no improvement with placebo. (46)
Preeclampsia
Endothelial dysfunction appears to be involved in the pathogenesis of preeclampsia. (47) In an animal model of experimental preeclampsia, IV administration of arginine (0.16 g/kg body weight/day) from gestational day 10 until term reversed hypertension, intrauterine growth retardation, proteinuria, and renal injury. (48) Intravenous infusion of arginine (30 g) in preeclamptic women has reportedly increased systemic NO production and reduced blood pressure. (49)
Human Immunodeficiency Virus (HIV) Infection and Acquired Immunodeficiency Syndrome (AIDS)
Arginine may be of benefit in individuals with HIV/AIDS. In a small pilot study of arginine supplementation in individuals with HIV, 11 patients were given 19.6 g/day arginine or placebo for 14 days. NK-cell cytotoxicity increased 18.9 lytic units, compared to an increase of 0.3 lytic units with placebo. This was not statistically significant, most likely due to the small number of patients in the study. (50)
A combination of glutamine, arginine, and hydroxymethylbutyrate (HMB) may prevent loss of lean body mass in individuals with AIDS cachexia. In a double-blind trial, AIDS patients with documented weight loss of at least five percent in the previous three months received either placebo or a combination of 3 g HMB, 14 g L-glutamine, and 14 g arginine given in two divided doses daily for eight weeks. At eight weeks, subjects consuming the mixture gained 3.0 [+ or -] 0.5 kg, while those supplemented with placebo gained only 0.37 [+ or -] 0.84 kg (p = 0.009). The weight gain in the supplemented group was predominately lean muscle mass, while the placebo group lost lean mass. (51)
A six-month, randomized, double-blind trial of an arginine/essential fatty acid combination was undertaken in patients with HIV. (52) Patients received a daily oral nutritional supplement (606 kcal supplemented with vitamins, minerals, and trace elements). In addition, half of the patients were randomized to receive 7.4 g arginine plus 1.7 g omega-3 fatty acids daily. Body weight increased similarly in both groups, and there was no change in immunological parameters. Clinical trials evaluating the effect of arginine as monotherapy for AIDS patients have yet to be conducted.
Growth Hormone Secretion and Athletic Performance
In rats, NO stimulates secretion of GH-releasing hormone (GHRH), thereby increasing secretion of GH. However, GHRH then increases production of NO in somatotroph cells, which subsequently inhibits GH secretion. In humans, arginine stimulates release of GH from the pituitary gland in some populations, but the mechanism is not well understood. Most studies suggest inhibition of somatostatin secretion is responsible for the effect. (53)
At high doses (approximately 250 mg/kg body weight), arginine aspartate increased GH secretion, (53) an effect of interest to body builders wishing to take advantage of the anabolic properties of the hormone. (54) In a controlled clinical trial, arginine and ornithine (500 mg of each, twice daily, five times per week) produced a significant decrease in body fat when combined with exercise. (55) Acute dosing of arginine (5 g taken 30 minutes before exercise) did not increase GH secretion, and may have impaired release of GH in young adults. (56) Longer-term, low-dose supplementation of arginine and ornithine (1 g each, five days per week for five weeks) resulted in higher gains in strength and enhancement of lean body mass, compared with controls receiving vitamin C and calcium. (57)
Growth hormone has been observed to be lower in older males than young men; however, data suggest oral arginine/lysine (3 g each daily) is not a practical means of enhancing long-term GH secretion in older men. (58)
Burns and Critical Trauma
Burn injuries significantly increase arginine oxidation and can result in depletion of arginine reserves. Total parenteral nutrition (TPN) increases conversion of arginine to ornithine and proportionally increases irreversible arginine oxidation, which, coupled with limited de novo synthesis from its immediate precursors, makes arginine conditionally essential in severely burned patients receiving TPN. (59) Several trials have demonstrated reduced length of hospital stay, fewer acquired infections, and improved immune function among burn (60) and trauma (61) patients supplemented with various combinations of fish or canola oil, nucleotides, and arginine.
Cancer
Animal research has shown large doses of arginine may interfere with tumor induction. (62) Short-term arginine supplementation may assist in maintenance of immune function during chemotherapy. Arginine supplementation (30 g/day for three days) reduced chemotherapy-induced suppression of lymphokine-activated killer cell cytotoxicity and lymphocyte mitogenic reactivity in patients with locally advanced breast cancer. (18,19) In another study, arginine supplementation (30 g/day for three days prior to surgery) significantly enhanced the activity of tumor-infiltrating lymphocytes in human colorectal cancers in vivo. (63) Arginine, RNA, and fish oil have been combined to improve immune function in cancer patients. (64-66)
On the other hand, arginine has also promoted cancer growth in animal and human research. (67) Polyamines act as growth factors for cancers. In several types of cancer, drugs are being investigated to inhibit ornithine decarboxylase (ODC), and hence inhibit polyamine formation. The possibility of arginine stimulating polyamine formation might be a concern in chronic administration, since both arginase and ODC appear to be up-regulated in some cancers.
Diabetes and Insulin Resistance Syndrome
Endothelium-dependent vascular relaxation is impaired in type 1 and type 2 diabetes mellitus (DM), and endothelial NO deficiency is a likely explanation. (68) Diabetes is associated with reduced plasma levels of arginine, (69) and evidence suggests arginine supplementation may be an effective way to improve endothelial function in individuals with diabetes. An IV bolus of 3-5 g arginine reduced blood pressure and platelet aggregation in patients with type 1 diabetes. (70) Low-dose IV arginine improved insulin sensitivity in obese patients and type 2 DM patients as well as in healthy subjects. (71) Arginine may also counteract lipid peroxidation and thereby reduce microangiopathic long-term complications of DM. (72)
After one week of oral arginine supplementation (9 g daily), 10 women with type 2 DM showed significant improvement in endothelial function, noted by a 50-percent increase in flow-mediated brachial dilation. (73)
A double-blind trial found oral arginine supplementation (3 g three times daily) significantly improved, but did not completely normalize, peripheral and hepatic insulin sensitivity in patients with type 2 diabetes. (74) In young patients with type 1 DM, however, oral arginine (7 g twice daily for six weeks) failed to improve endothelial function. (75)
Gastrointestinal Conditions
Gastritis and Ulcer
Preliminary evidence suggests arginine accelerates ulcer healing due to its hyperemic, angiogenic, and growth-promoting actions, possibly involving NO, gastrin, and polyamines. (76,77) No clinical trials have yet explored the efficacy of arginine supplementation as a treatment for gastritis or peptic ulcer in humans.
Gastroesophageal Reflux (GERD) and Sphincter Motility Disorders
A small, double-blind trial found oral arginine supplementation significantly decreased the frequency and intensity of chest pain attacks, as well as the number of nitroglycerin tablets taken for analgesia, in patients with esophageal motility disorders. (78) However, in another study, arginine infusions (500 mg/kg body weight/120 min) failed to affect lower esophageal sphincter motility. (79) No studies have yet explored the efficacy of arginine supplements for GERD.
Genitourinary Conditions Erectile Dysfunction (ED)
In a small, uncontrolled trial, men with ED were given 2.8 g arginine daily for two weeks. Forty percent of men in the treatment group experienced improvement, compared to none in the placebo group. (80) In a larger double-blind trial, men with ED were given 1,670 mg arginine daily or a matching placebo for six weeks. (81) Arginine supplementation was effective at improving ED in men with abnormal nitric oxide metabolism. However, another double-blind trial of arginine for ED (500 mg three times daily for 17 days) found the amino acid no more effective than placebo. (82)
Infertility, Female
Supplementation with oral arginine (16 g/ day) in poor responders to in vitro fertilization improved ovarian response, endometrial receptivity, and pregnancy rate in one study. (83)
Infertility, Male
Arginine is required for normal spermatogenesis. Over 50 years ago, researchers found that feeding an arginine-deficient diet to adult men for nine days decreased sperm counts by approximately 90 percent and increased the percentage of non-motile sperm approximately 10-fold. (84) Oral administration of 500 mg arginine-HCl per day to infertile men for 6-8 weeks markedly increased sperm count and motility in a majority of patients, and resulted in successful pregnancies. (85) Similar effects on oligospermia and conception rates have been reported in other preliminary trials. (86-89) However, when baseline sperm counts were less than 10 million/mL, arginine supplementation produced little or no improvement. (90,91)
Interstitial Cystitis (IC)
In an uncontrolled trial, 10 patients with IC took 1.5 g arginine daily for six months. Supplementation resulted in a significant decrease in urinary voiding discomfort, lower abdominal pain, and vaginal/urethral pain. Urinary frequency during the day and night also significantly decreased. (92) In a five-week uncontrolled trial, however, arginine supplementation was not effective, even at higher doses of 3-10 g daily. (93) In a randomized, double-blind trial of arginine for IC, patients took 1.5 g arginine daily for three months. Twenty-nine percent of patients in the arginine group and eight percent in the placebo group experienced clinical improvement (i.e., decreased pain and urgency) by the end of the trial (p = 0.07). The results fell short of statistical significance, most likely because of the small sample size (n = 53).
Perioperative Nutrition
Arginine is a potent immunomodulator. Evidence is mounting for a beneficial effect of arginine supplementation in catabolic conditions such as sepsis and postoperative stress. Supplemental arginine appears to up-regulate immune function and reduce the incidence of postoperative infection. (94) Two controlled trials have demonstrated increased lymphocyte mitogenesis and improved wound healing in experimental surgical wounds in volunteers given 17-25 g oral arginine daily. (95,96) Similar results have been obtained in healthy elderly volunteers. (97)
Preterm Labor and Delivery
Evidence from human and animal studies indicates nitric oxide inhibits uterine contractility and may help maintain uterine quiescence during pregnancy. (98) IV arginine infusion (30 g over 30 min) in women with premature uterine contractions transiently reduced uterine contractility. (99) Further research is needed to confirm the efficacy and safety of arginine in prevention of preterm delivery.
Senile Dementia
Arginine (1.6 g/day) in 16 elderly patients with senile dementia reduced lipid peroxidation and increased cognitive function. (100)
Side Effects and Toxicity
Significant adverse effects have not been observed with arginine supplementation. People with renal failure or hepatic disease may be unable to appropriately metabolize and excrete supplemental arginine and should be closely monitored when taking arginine supplements.
Dosage
Doses of arginine used in clinical research have varied considerably, from as little as 500 mg/day for oligospermia to as much as 30 g/day for cancer, preeclampsia, and premature uterine contractions. Typical daily doses fall into either the 1-3 g or 7-15 g range, depending on the condition being treated. Because of the pharmacokinetics of L-arginine, use of a sustained-release preparation may be preferable, in order to keep blood levels more constant over time.
Warnings and Contraindications
It has been postulated, on the basis of older in vitro data (101) and anecdotal reporting, that arginine supplementation might be contraindicated in persons with herpes infections (i.e., cold sores, genital herpes). The assumption is that arginine might stimulate replication of the virus and/or provoke an outbreak; however, this caution has not been validated by controlled clinical trials.
Bronchoconstriction is reportedly inhibited by the formation of NO in the airways of asthmatic patients, and a bronchoprotective effect of NO in asthma has been proposed. (102) Airway obstruction in asthma might be associated with endogenous NO deficiency caused by limited availability of NO synthase substrate (i.e., arginine). However, oral arginine (50 mg/kg body weight) in asthmatic patients triggered by a histamine challenge produced only a marginal, statistically insignificant improvement of airway hyper-responsiveness to histamine. (103) In fact, it is unclear whether NO acts as a protective or a stimulatory factor in airway hyper-responsiveness.
Since polyamines act as growth factors for cancers, and arginine may stimulate polyamine synthesis, chronic administration of arginine in cancer patients should probably be avoided until information arises regarding the safety of this practice.
References
(1.) Castillo L, Chapman TE, Sanchez M, et al. Plasma arginine and citrulline kinetics in adults given adequate and arginine-free diets. Proc Natl Acad Sci USA 1993;90:7749-7753.
(2.) Castillo L, Ajami A, Branch S, et al. Plasma arginine kinetics in adult man: response to an arginine-free diet. Metabolism 1994;43:114-122.
(3.) Abcouwer SF, Souba WW. Glutamine and arginine. In: Shils ME, Olson JA, Shike M, Ross AC, eds. Modern Nutrition in Health and Disease, 9 ed. Baltimore, MD: Williams & Wilkins; 1999:559-569.
(4.) Wu G, Davis PK, Flynn NE, et al. Endogenous synthesis of arginine plays an important role in maintaining arginine homeostasis in postweaning growing pigs. J Nutr 1997;127:2342-2349.
(5.) Dhanakoti SN, Brosnan JT, Herzberg GR, Brosnan ME. Renal arginine synthesis: studies in vitro and in vivo. Am J Physiol 1990;259:E437-E442.
(6.) Preiser JC, Berre PJ, Van Gossum A, et al. Metabolic effects of arginine addition to the enteral feeding of critically ill patients. JPEN J Parenter Enteral Nutr 2001;25:182-187.
(7.) Castillo L, Sanchez M, Vogt J, et al. Plasma arginine, citrulline, and ornithine kinetics in adults, with observations on nitric oxide synthesis. Am J Physiol 1995;268:E360-E367.
(8.) Wu G, Morris SM Jr. Arginine metabolism: nitric oxide and beyond. Biochem J 1998;336:1-17.
(9.) Meijer AJ, Lamers WH, Chamuleau RA. Nitrogen metabolism and ornithine cycle function. Physiol Rev 1990;70:701-748.
(10.) Wu G, Meininger CJ. Arginine nutrition and cardiovascular function. J Nutr 2000;130:2626-2629.
(11.) Gross SS, Wolin MS. Nitric oxide: pathophysiological mechanisms. Annu Rev Physiol 1995;57:737-769.
(12.) Wink DA, Hanbauer I, Grisham MB, et al. Chemical biology of nitric oxide: regulation and protective and toxic mechanisms. Curr Top Cell Regul 1996;34:159-187.
(13.) Umans JG, Levi R. Nitric oxide in the regulation of blood flow and arterial pressure. Annu Rev Physiol 1995;57:771-790.
(14.) Hare JM, Colucci WS. Role of nitric oxide in the regulation of myocardial function. Prog Cardiovasc Dis 1995;38:155-166.
(15.) Lyons CR. The role of nitric oxide in inflammation. Adv Immunol 1995;60:323-371.
(16.) Brune B, Messmer UK, Sandau K. The role of nitric oxide in cell injury. Toxicol Lett 1995;82-83:233-237.
(17.) Wink DA, Cook JA, Pacelli R, et al. Nitric oxide (NO) protects against cellular damage by reactive oxygen species. Toxicol Lett 1995;82-83:221-226.
(18.) Brittenden J, Heys SD, Ross J, et al. Natural cytotoxicity in breast cancer patients receiving neoadjuvant chemotherapy: effects of L-arginine supplementation. Eur J Surg Oncol 1994;20:467-472.
(19.) Brittenden J, Park KGM, Heys SD, et al. L-arginine stimulates host defenses in patients with breast cancer. Surgery 1994;115:205-212.
(20.) Imms FJ, London DR, Neame RL. The secretion of catecholamines from the adrenal gland following arginine infusion in the rat. J Physiol 1969;200:55P-56P.
(21.) Palmer JP, Walter RM, Ensinck JW. Arginine-stimulated acute phase of insulin and glucagon secretion. I. In normal man. Diabetes 1975;24:735-740.
(22.) Rakoff JS, Siler TM, Sinha YN, Yen SS. Prolactin and growth hormone release in response to sequential stimulation by arginine and synthetic TRE J Clin Endocrinol Metab 1973;37:641-644.
(23.) Knopf RF, Conn JW, Fajans SS, et al. Plasma growth hormone response to intravenous administration of amino acids. J Clin Endocrinol Metab 1965;25:1140-1144.
(24.) Merimee TJ, Lillicrap DA, Rabinowitz D. Effect of arginine on serum-levels of human growth-hormone. Lancet 1965;2:668-670.
(25.) Lekakis J, Papathanasieu S, Papamicheel C, et al. Oral 1-arginine improves endothelial dysfunction in patients with essential hypertension. J Am Coll Cardiol 2001:260A.
(26.) Boger GI, Maas R, Schwedhelm E, et al. Improvement of endothelium-dependent vasodilation by simvastatin is potentiated by combination with 1-arginine in patients with elevated asymmetric dimethylarginine levels. J Am Coll Cardiol 2004:525A.
(27.) Boger RH, Vallance P, Cooke JR Asymmetric dimethylarginine (ADMA): a key regulator of nitric oxide synthase. Atherosclerosis Suppl 2003;4:1-3.
(28.) Boger RH. Asymmetric dimethylarginine, an endogenous inhibitor of nitric oxide synthase, explains the "L-arginine paradox" and acts as a novel cardiovascular risk factor. J Nutr 2004; 134:2842S-2847S.
(29.) Boger RH, Ron ES. L-Arginine improves vascular function by overcoming deleterious effects of ADMA, a novel cardiovascular risk factor. Altern Med Rev 2005;10:14-23.
(30.) Maxwell AJ, Zapien MP, BS, Pearce GL, et al. Randomized trial of a medical food for the dietary management of chronic, stable angina. J Am Coll Cardiol 2002;39:37-45.
(31.) Blum A, Porat R, Rosenschein U, et al. Clinical and inflammatory effects of dietary L-arginine in patients with intractable angina pectoris. Am J Cardiol 1999;83:1488-1490.
(32.) Ceremuzynski L, Chamiec T, Herbaczynska-Cedro K. Effect of supplemental oral L-arginine on exercise capacity in patients with stable angina pectoris. Am J Cardiol 1997;80:331-333.
(33.) Walker HA, McGing E, Fisher I, et al. Endothelium-dependent vasodilation is independent of the plasma L-arginine/ADMA ratio in men with stable angina: lack of effect of oral L-arginine on endothelial function, oxidative stress and exercise performance. J Am Coll Cardiol 2001;38:499-505.
(34.) Bednarz B, Wolk R, Chamiec T, et al. Effects of oral L-arginine supplementation on exercise-induced QT dispersion and exercise tolerance in stable angina pectoris. Int J Cardiol 2000;75:205-210.
(35.) Egashira K, Hirooka Y, Kuga T, et al. Effects of L-arginine supplementation on endothelium-dependent coronary vasodilation in patients with angina pectoris and normal coronary arteriograms. Circulation 1996;94:130-134.
(36.) Rector TS, Bank A, Mullen KA, et al. Randomized, double-blind, placebo controlled study of supplemental oral L-arginine in patients with heart failure. Circulation 1996;93:2135-2141.
(37.) Watanabe G, Tomiyama H, Doba N. Effects of oral administration of L-arginine on renal function in patients with heart failure. J Hypertens 2000;18:229-234.
(38.) Yousufuddin M, Flather M, Shamim W, et al. A short course of L-arginine improves exercise capacity and endothelial function in chronic heart failure: a prospective, randomised, double blind trial. J Am Coll Cardiol 2001:211A.
(39.) Houghton JL, Toresoff MT, Kuhner PA, et al. African American race predicts improvement in coronary microvascular endothelial function after L-arginine. J Am Coll Cardiol 2001:258A.
(40.) Sanders PW. Salt-sensitive hypertension: lessons from animal models. Am J Kidney Dis 1996;28:775-782.
(41.) Calver A, Collier J, Vallance P. Dilator actions of arginine in human peripheral vasculature. Clin Sci 1991;81:695-700.
(42.) Pezza V, Bernardini F, Pezza E, et al. Study of supplemental oral L-arginine in hypertensives treated with enalapril + hydrochlorothiazide. Am J Hypertens 1998;11:1267-1270.
(43.) Siani A, Pagano E, Iacone R, et al. Blood pressure and metabolic changes during dietary L-arginine supplementation in humans. Am J Hypertens 2000;13:547-551.
(44.) Maccario M, Oleandri SE, Procopio M, et al. Comparison among the effects of arginine, a nitric oxide precursor, isosorbide dinitrate and molsidomine, two nitric oxide donors, on hormonal secretions and blood pressure in man. J Endocrinol Invest 1997;20:488-492.
(45.) Schulze-Neick I, Penny DJ, Rigby ML, et al. L-arginine and substance P reverse the pulmonary endothelial dysfunction caused by congenital heart surgery. Circulation 1999;100:749-755.
(46.) Boger RH, Bode-Boger SM, Thiele W, et al. Restoring vascular nitric oxide formation by L-arginine improves the symptoms of intermittent claudication in patients with peripheral arterial occlusive disease. J Am Coll Cardiol 1998;32:1336-1344.
(47.) Roberts JM. Objective evidence of endothelial dysfunction in preeclampsia. Am J Kidney Dis 1999;33:992-997.
(48.) Helmbrecht GD, Farhat MY, Lochbaum L, et al. L-arginine reverses the adverse pregnancy changes induced by nitric oxide synthase inhibition in the rat. Am J Obstet Gynecol 1996;175:800-805.
(49.) Facchinetti F, Longo M, Piccinini F, et al. L-arginine infusion reduces blood pressure in preeclamptic women through nitric oxide release. J Soc Gynecol Invest 1999;6:202-207.
(50.) Swanson B, Keithley JK, Zeller JM, Sha BE. A pilot study of the safety and efficacy of supplemental arginine to enhance immune function in persons with HIV/AIDS. Nutrition 2002;18:688-690.
(51.) Clark RH, Feleke G, Din M, et al. Nutritional treatment for acquired immunodeficiency virus-associated wasting using beta-hydroxy beta-methylbutyrate, glutamine, and arginine: a randomized, double-blind, placebo-controlled study. JPEN J Parenter Enteral Nutr 2000;24:133-139.
(52.) Pichard C, Sudre P, Karsegard V, et al. A randomized double-blind controlled study of 6 months of oral nutritional supplementation with arginine and omega-3 fatty acids in HIV-infected patients. Swiss HIV Cohort Study. AIDS 1998;12:53-63.
(53.) Besset A, Bonardet A, Rondouin G, et al. Increase in sleep related GH and Pr1 secretion after chronic arginine aspartate administration in man. Acta Endocrinol 1982;99:18-23.
(54.) Macintyre JG. Growth hormone and athletes. Sports Med 1987;4:129-142.
(55.) Elam RE Morphological changes in adult males from resistance exercise and amino acid supplementation. J Sports Med Phys Fitness 1988;28:35-39.
(56.) Marcell TJ, Taaffe DR, Hawkins SA, et al. Oral arginine does not stimulate basal or augment exercise-induced GH secretion in either young or old adults. J Gerontol A Biol Sci Med Sci 1999;54: M395-M399.
(57.) Elam RP. Effect of arginine and ornithine on strength, lean body mass and urinary hydroxyproline in adult males. J Sports Nutr 1989;29:52-56.
(58.) Corpas E, Blackman MR, Roberson R, et al. Oral arginine-lysine does not increase growth hormone or insulin-like growth factor-I in old men. J Gerontol 1993;48:M128-M133.
(59.) Yu YM, Ryan CM, Castillo L, et al. Arginine and ornithine kinetics in severely burned patients: increased rate of arginine disposal. Am J Physiol Endocrinol Metab 2001;280:E509-E517.
(60.) Bower RH, Cerra FB, Bershadsky B, et al. Early enteral administration of a formula (Impact) supplemented with arginine, nucleotides, and fish oil in intensive care unit patients: results of a multicenter, prospective, randomized clinical trial. Crit Care Med 1995;23:436-439.
(61.) Weimann A, Bastian L, Bischoff WE, et al. Influence of arginine, omega-3 fatty acids and nucleotide-supplemented enteral support on systemic inflammatory response syndrome and multiple organ failure in patients after severe trauma. Nutrition 1998;14:165-172.
(62.) Takeda Y, Tominga T, Tei N, et al. Inhibitory effect of L-arginine on growth of rat mammary tumors induced by 7, 12, dimethlybenz(a)anthracine. Cancer Res 1975;35:390-393.
(63.) Heys SD, Segar A, Payne S, et al. Dietary supplementation with L-arginine: modulation of tumour-infiltrating lymphocytes in patients with colorectal cancer. Br J Surg 1997;84:238-241.
(64.) Kemen M, Senkal M, Homann HH, et al. Early postoperative enteral nutrition with arginine-omega-3 fatty acids and ribonucleic acid-supplemented diet versus placebo in cancer patients: an immunologic evaluation of Impact. Crit Care Med 1995;23:652-659.
(65.) Gianotti L, Braga M, Fortis C, et al. A prospective, randomized clinical trial on perioperative feeding with an arginine-, omega-3 fatty acid-, and RNA-enriched enteral diet: effect on host response and nutritional status. JPEN J Parenter Enteral Nutr 1999;23:314-320.
(66.) van Bokhorst-De Van Der Schueren MA, Quak JJ, von Blomberg-van der Flier BM, et al. Effect of perioperative nutrition, with and without arginine supplementation, on nutritional status, immune function, postoperative morbidity, and survival in severely malnourished head and neck cancer patients. Am J Clin Nutr 2001;73:323-332.
(67.) Park KGM. The Sir David Cuthbertson Medal Lecture 1992. The immunological and metabolic effects of L-arginine in human cancer. Proc Nutr Soc 1993;52:387-401.
(68.) Pieper GM. Review of alterations in endothelial nitric oxide production in diabetes. Hypertension 1998;31:1047-1060.
(69.) Pieper GM, Siebeneich W, Dondlinger LA. Short-term oral administration of L-arginine reverses defective endothelium-dependent relaxation and cGMP generation in diabetes. Eur J Pharmacol 1996;317:317-320.
(70.) Giugliano D, Marfella R, Verrazzo G, et al. L-arginine for testing endothelium-dependent vascular functions in health and disease. Am J Physiol 1997;273:E606-E612.
(71.) Wascher TC, Graier WF, Dittrich P, et al. Effects of low-dose L-arginine on insulin-mediated vasodilatation and insulin sensitivity. Eur J Clin Invest 1997;27:690-695.
(72.) Lubec B, Hayn M, Kitzmuller E, et al. L-arginine reduces lipid peroxidation in patients with diabetes mellitus. Free Radic Biol Med 1997;22:355-357.
(73.) Regensteiner JG, Popylisen S, Bauer TA, et al. Oral 1-arginine and vitamins E and C improve endothelial function in women with type 2 diabetes. Vasc Med 2003; 8:169-175.
(74.) Piatti PM, Monti LD, Valsecchi G, et al. Long-term oral L-arginine administration improves peripheral and hepatic insulin sensitivity in type 2 diabetic patients. Diabetes Care 2001;24:875-880.
(75.) Mullen MJ, Wright D, Donald AE, et al. Atorvastatin but not L-arginine improves endothelial function in type I diabetes mellitus: a double-blind study. J Am Coll Cardiol 2000;36:410-416.
(76.) Brzozowski T, Konturek SJ, Sliwowski Z, et al. Role of L-arginine, a substrate for nitric oxidesynthase, in gastroprotection and ulcer healing. J Gastroenterol 1997;32:442-452.
(77.) Brzozowski T, Konturek SJ, Drozdowicz D, et al. Healing of chronic gastric ulcerations by Larginine. Role of nitric oxide, prostaglandins, gastrin and polyamines. Digestion 1995;56:463471.
(78.) Bortolotti M, Brunelli F, Sarti P, Miglioli M. Clinical and manometric effects of L-arginine in patients with chest pain and oesophageal motor disorders, Ital J Gastroenterol Hepatol 1997;29:320-324.
(79.) Straathof JW, Adamse M, Onkenhout W, et al. Effect of L-arginine on lower oesophageal sphincter motility in man. Eur J Gastroenterol Hepatol 2000;12:419-424.
(80.) Zorgniotti AW, Lizza EF. Effect of large doses of the nitric oxide precursor, L-arginine, on erectile dysfunction. Int J Impot Res 1994;6:33-36.
(81.) Chen J, Wollman Y, Chernichovsky T, et al. Effect of oral administration of high-dose nitric oxide donor L-arginine in men with organic erectile dysfunction: results of a double-blind, randomized study. BJU Int 1999;83:269-273.
(82.) Klotz T, Mathers MJ, Braun M, et al. Effectiveness of oral L-arginine in first-line treatment of erectile dysfunction in a controlled crossover study. Urol Int 1999;63:220-223.
(83.) Battaglia C, Salvatori M, Maxia N, et al. Adjuvant L-arginine treatment for in vitro fertilization in poor responder patients. Hum Reprod 1999;14:1690-1697.
(84.) Holt LE Jr, Albanese AA. Observations on amino acid deficiencies in man. Trans Assoc Am Physicians 1944;58:143-156.
(85.) Tanimura J. Studies on arginine in human semen. Part II. The effects of medication with L-arginineHC1 on male infertility. Bull Osaka Med School 1967;13:84-89.
(86.) De Aloysio D, Mantuano R, Mauloni M, Nicoletti G. The clinical use of arginine aspartate in male infertility. Acta Eur Fertil 1982;13:133-167.
(87.) Scibona M, Meschini P, Capparelli S, et al. Larginine and male infertility. Minerva Urol Nefrol 1994;46:251-253.
(88.) Schacter A, Goldman JA, Zukerman Z. Treatment of oligospermia with the amino acid arginine. J Urol 1973;110:311-313.
(89.) Schacter A, Friedman S, Goldman JA, Eckerling B. Treatment of oligospermia with the amino acid arginine. Int J Gynaecol Obstet 1973;11:206-209.
(90.) Pryor JP, Blandy JP, Evans P, et al. Controlled clinical trial of arginine for infertile men with oligozoospermia. Br J Urol 1978;50:47-50.
(91.) Mroueh A. Effect of arginine on oligospermia. Fertil Steril 1970;21:217-219.
(92.) Smith SD, Wheeler MA, Foster HE Jr, Weiss RM. Improvement in interstitial cystitis symptom scores during treatment with oral L-arginine. J Urol 1997;158:703-708.
(93.) Ehren I, Lundberg JO, Adolfsson J. Effects of L-arginine treatment on symptoms and bladder nitric oxide levels in patients with interstitial cystitis. Urology 1998;52:1026-1029.
(94.) Evoy D, Lieberman MD, Fahey TJ 3rd, Daly JM. Immunonutrition: the role of arginine. Nutrition 1998;14:611-617.
(95.) Barbul A, Rettura G, Levenson SM, et al. Wound healing and thymotropic effects of arginine: a pituitary mechanism of action. Am J Clin Nutr 1983;37:786-794.
(96.) Barbul A, Lazarou SA, Efron DT, et al. Arginine enhances wound healing and lymphocyte immune responses in humans. Surgery 1990;108:331-337.
(97.) Kirk SJ, Hurson M, Regan MC, et al. Arginine stimulates wound healing and immune function in elderly human beings. Surgery 1993; 114:155-160.
(98.) Buhimschi IA, Saade GR, Chwalisz K, Garfield RE. The nitric oxide pathway in pre-eclampsia: pathophysiological implications. Human Reprod Update 1998;4:25-42.
(99.) Facchinetti F, Neri I, Genazzani AR. L-arginine infusion reduces preterm uterine contractions. J Perinat Med 1996;24:283-285.
(100.) Ohtsuka Y, Nakaya J. Effect of oral administration of L-arginine on senile dementia. Am J Med 2000; 108:439.
(101.) Tankersley RW. Amino acid requirements of herpes simplex virus in human cells. J Bacteriol 1964;87:609-613.
(102.) Ricciardolo FL, Geppetti P, Mistretta A, et al. Randomised double-blind placebo-controlled study of the effect of inhibition of nitric oxide synthesis in bradykinin-induced asthma. Lancet 1996;348:374-377.
(103.) de Gouw HW, Verbruggen MB, Twiss IM, Sterk PJ. Effect of oral L-arginine on airway hyperresponsiveness to histamine in asthma. Thorax 1999;54:1033-1035.
COPYRIGHT 2005 Thorne Research Inc.
COPYRIGHT 2005 Gale Group