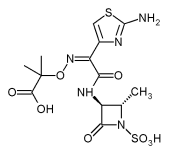
Sir,
Pseudomonas aeruginosa exhibits resistance to a variety of antimicrobials including beta lactams. Carbapenems are often used as antibiotics for treatment of infections caused by β-lactam resistant P. aeruginosa1. However a group of carbapenem hydrolyzing beta-lactamases known as metallo beta-lactamases (MBLs), belonging to Amblers' class B2 or to Bush et al group 3(3) classification of ESBLs (extended spectrum beta-lactamases), have been reported in several countries, particularly in clinical isolates of P. aeruginosa4-6. The MBLs are inhibited in vitro by CuCl^sub 3^, FeCl^sub 3^, EDTA, sodium mercaptoacetic acid (SMA), 2 mercaptopropionic acid (2MPA) and 2-mercaptoethanol (2ME)7 but not by clavulanic acid, sulbactam or tazobactum8,9. Aztreonam is the treatment of choice for infections with organisms producing MBLs3. In order to select the correct antibiotic for treatment and to present dissemination of such infection, it is important to detect these strains in the laboratory.
We studied 174 P. aeruginosa isolates obtained from various clinical specimens received in the Department of Microbiology, Mahatma Gandhi Institute of Medical Sciences, Sevagram between January and August 2004 and identified as per standard techniques. Antibiotic susceptibility to chloramphenicol (30 µg), gentamycin (10 µg), amikacin (10 µg), ceftriáxone (30 µg), ciprofloxacin (10 µg), ceftazidine(30 µg) and imipenem (10 µg) was performed by Kirby Bauer's disk diffusion method as per National Committee for Clinical Laboratory Standards (NCCLS) guidelines (2002)10. E. coli ATCC 25922 and P. aeruginosa ATCC 27853 (courtesy Microbiology Department, Lady Hardinge Medical College, New Delhi) were used as negative controls. Isolates resistant to imipenem (IMP) and ceftazidime (CAZ) were suspected to be MBL producers and were confirmed by double disk synergy test (DDST)7.
In brief, a colony of the suspected isolate was suspended in Muller Hinton broth and diluted to obtain 10^sup 6^ cfu/ml. Lawn culture was made on Muller Hinton agar and DDST was put. The combinations used in DDST were: CAZ-2ME, CAZ-EDTA, IMP-2ME and IMP-EDTA. All MBL positives were checked twice for reproducibility.
All MBL positive isolates were also subjected to DDST using ceftazidime-amoxyclav and cefaperazone-sulbactam combination to determine if the MBLs were inhibited by clavulanic acid or sulbactam.
Of the 174 isolates studied, 15 (8.62%) were suspected to be MBL producers (resistant to both IMP and CAZ). In DDST, all 15 suspected isolates gave a clear large zone of inhibition around CAZ disc towards EDTA as well as 2ME (Table and Fig.). When IMP was used in place of CAZ, only 14 of the 15 isolates could be confirmed for MBL production and 3 of these gave small zone with EDTA were as with 2ME 11 gave small and 3 narrow zone of inhibition (Table; Fig.) in contrast to all 15 giving large zone when CAZ was used with these inhibitors.
MBLs are enzymes that are either chromosomal or plasmid encoded4,5,11,12. IMP-1 was the first MBL identified in P. aeruginosa12-13. Several other novel MBLs like VIM-1, VIM-2 and VIM-3(5,6) have been identified recently from P. aeruginosa.
A preliminary study from an urban hospital in Bangalore14 reported 6 of the 50 isolates of P. aeruginosa to be MBL positive. Our study revealed that MBL producing P. aeruginosa is prevalent in this rural hospital also. All our MBL producer isolates were resistant to all antibiotics tested and the MBL production was not inhibited by beta lactam inhibitors (clavulanic acid and sulbactam) as has been reported earlier8,9,14,15.
In our study DDST using CAZ with either 2ME or EDTA gave better results than IMP-2ME or IMP-EDTA. CAZ was found to be the most useful substrate for detection of IMP-18 or both IMP-1 and VIM producers6 by DDST. However, IMP disk and a disk impregnated with both EDTA (750 µg) + SMA (2 µg)1 or a single disc containing both IMP and EDTA (750 µg)16 were found to be excellent in differentiating MBL producers.
Different methods such as PCR13, E test17, microdilution test18 and modified Hodge19 test have been used besides DDST to identify MBL producers. However, these test are either not cost-effective (PCR, E test) or time consuming (microdilution) for routine testing in clinical laboratories. PCR has become more difficult with the increase in number of types of MBLs16. Hodge test performed on positive isolates in our laboratory did not give an appreciable distorted zone of inhibition and therefore was not found suitable when compared to DDST (unpublished data).
Considering the need to institute correct antibiotics to the patients infected with MBL producers and to prevent spread of MBL positive organisms, all clinical microbiology laboratories must routinely identify the MBL producers, and simple DDST test using CAZ-2ME or CAZ-EDTA could be used in developing countries.
References
1. Lee K, Lim YS, Yong D, Yum JH, Chong Y. Evaluation of the Hedge test and the imipenem-EDTA double-disk synergy test for differentiating metallo-β-lactamase-producing isolates of Pseudomonas spp. and Acinetobacter spp. J Clin Microbiol 2003; 41 : 4623-9.
2. Ambler RP. The structure of β-lactamases. Philos Trans R Soc Land B Biol Sci 1980; 289 : 321-31.
3. Bush K, Jacoby GA, Medeiros AA. A functional classification scheme for β-lactamases and its correlation with molecular structure. Antimicrob Agents Chemother 1995; 39 : 1211-33.
4. Lauretti L, Riccio ML, Mazzariol A, Cornaglia G, Amicosante G, Fontana R, et al. Cloning and characterization of Bla^sub VIM^, a nem integron- borne metallo-β-lactamase gene from a Pseudomonas aeruginosa clinical isolate. Antimicrob Agent Chemother 1999; 43 : 1584-90.
5. Poirel I, Naas T, Nicolas D, Collet L, Bellais S, Cavallo JD, et al. Characterisation of VIM-2, acarbapenem hydrolyzing metallo-β-lactamase and its plasmid and interon-borne gene from a Pseudomonas aeruginosa clinical isolates in France. Antimicrob Agent Chemother 1999; 44: 891-7.
6. Yan JJ, Hsueb PR, Ko WC, Luh KT, Tsai SH, Wu HM, et al. Metallo-β-lactamase in clinical Pseudomonas isolates in Taiwan and identification of VIM-3, a novel variant of the VIM-2 enzyme. Antimicrob Agent Chemother 2001; 45 : 2224-8.
7. Arakawa Y, Shibata N, Shibayama K, Kurokawa H, Yagi T. Fujiwara H, et al. Convenient test for screening metallo-β-lactamase - producing Gram-negative bacteria by using thiol compounds. J Clin Microbiol 2000; 38 : 40-3.
8. Bush K, Macalintal C, Rasmussen BA, Lee VJ, Yang Y. Kinetic interactions of tazobactam with β-lactamases from all major structurel classes. Antimicrob Agent Chemother 1993; 37: 851-8.
9. Ohsuka S, Arakawa Y, Horii T, Ito H, Ohta M. Effect of pH on activities of novel β-lactamases and β-lactamase inhibitors against these β-lactamases. Antimicrob Agent Chemother 1991; 39: 1856-8.
10. Performance standards for antimicrobial susceptibility testing. Twelfth informational supplement. NCCLS document M100-S12 Vol.22. No.1 (ISBN 1-56238-454-6), NCCLS, 940 West Valley Road, Suite 1400, Wayne, Pennsylvania 1987-1988 USA, 2002.
11. Hancock REW. Resistance mechanisms in Pseudomonas aeruginosa and other non fermentative Gram negative bacteria. Clin Infect Dis 1998; 27 (Suppl 1): S93-9.
12. Senda K, Arakawa Y, Nakashima K, Ito H, Ichiyama S, Shimokata K, et al. Multifocal outbreaks of metallo -β-lactamase producing Pseudomonas aeruginosa resistant to broad-spectrum β-lactams, including carbapenems. Antimicrob Agent Chemother 1996; 40 : 349-53.
13. Senda K, Arakawa Y, Ichiyama S, Nakashuma K, Ito H, Ohsuka S, et al. PCR detection of metallo- β-lactamases gene (bla ^sub IMP^) in Gram-negative rods resistant to broad spectrum β-lactams. J Clin Microbiol 1996; 34 : 2909-13.
14. Navaneeth BV, Sridaran D, Sahay D, Belwadi MR. A preliminary study on metallo- β-lactamase producing Pseudomonas aeruginosa in hospitalized patients. Indian J Med Res 2002; 116: 264-7.
15. Bush K. Metallo- β-lactamase - a class apart. Clin Infect Dis 1998; 27 (Suppl 1): 548-53.
16. Yong D, Lee K, Yum JH, Shin HB, Rossolini GM, Chong Y. Imipenem-EDTA disk method for differentiation of metalloβ-lactamases producing clinical isolates of Pseudomonas spp. and Acinetobacter spp. J Clin Microbiol 2002; 40 : 3798-801.
17. Walsh TR, Bolmstrom A, Qwarnstrom A, Gales A. Evaluation of a new E test for detecting metallo-β-lactamase in routine clinical testing. J Clin Microbiol 2002; 40 : 2755-9.
18. Migliavacca R, Docquier JD, Mugnaioli C, Amicosante G, Daturi R, Lee K, et al. Simple microdilution test for detection of metallo- β-lactamase production in Pseudomonas aeruginosa. J Clin Microbiol 2002; 40 : 4388-90.
19. Lee K, Chong Y, Shin HB, Kim YA, Yong D, Yum JH. Modified Hodge test EDTA disk synergy tests to screen metallo- β-lactamase-producing strains of Pseudomonas and Acinetobacter species. Clin Microbiol Infect 2001; 7 : 88-91.
D.K. Mendiratta, Vijayshri Deotale & Pratibha Narang
Department of Microbiology
Mahatma Gandhi Institute of Medical Sciences
Sevagram 442102 Maharashtra, India
e-mail: dkmendiratta@rediffmail.com
Copyright Indian Council of Medical Research May 2005
Provided by ProQuest Information and Learning Company. All rights Reserved