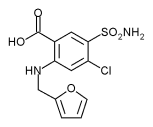
The aim of this study is to investigate the effects of inhaled furosemide on the sensation of dyspnea produced during exercise in patients with stable chronic obstructive pulmonary disease (COPD). In a double-blind, randomized, crossover study we compared the effect of inhaled furosemide on dyspneic sensation during exercise testing with that of placebo. Spirometry and incremental and constant-load exercise testing were performed after inhalation of placebo or furosemide on 2 separate days in 19 patients with moderate or severe COPD. Subjects were asked to rate their sensation of respiratory discomfort using a 100-mm visual analog scale. There was significant improvement in mean FEV^sub 1^ and FVC after inhalation of furosemide (p = 0.038 and 0.005, respectively) but not after placebo. At standardized exercise time during constant-load exercise testing but not during incremental exercise, the mean dyspneic visual analog scale score was lower after inhalation of furosemide compared with placebo (33.7 ± 25.2 vs. 42.4 ± 24.0 mm, respectively, p = 0.014). We conclude that inhalation of furosemide alleviates the sensation of dyspnea induced by constant-load exercise testing in patients with COPD and that there is significant bronchodilation after inhalation of furosemide compared with placebo in these patients.
Keywords: exercise capacity; bronchodilation; breathlessness; exercise test; spirometry
Dyspnea is the hallmark symptom of chronic obstructive pulmonary disease (COPD) and is a major cause of disability and anxiety associated with the disease (1). It is also the reason most patients with COPD seek medical attention (1). Patients with COPD are known to experience dyspnea at lower levels of exercise than unaffected individuals as a consequence of multiple pathophysiologic factors (2). Although exertional symptoms may be mild at the outset, exercise limitation is the most disabling and distressing consequence of COPD for the majority of patients.
The interest in the clinical application of inhaled furosemide has grown in the last few years. Inhalation of furosemide has been shown to have an inhibitory effect on experimentally induced cough (3) and to prevent bronchoconstriction in patients with asthma (4-6). Recently, inhaled furosemide has been observed to decrease the sensation of experimentally induced dyspnea. In a double-blind, randomized, crossover study (7) performed on 12 healthy subjects, there was remarkable prolongation of both the total breathholding time and the period of breathhold with no respiratory sensation after inhalation of furosemide. The development of respiratory discomfort induced by a combination of resistive loading and hypercapnia was also much slower and less than that observed after inhalation of placebo.
Because patients with COPD may experience, especially during exercise, dyspnea similar to that experienced by the normal subjects with resistive loading and hypercapnia in the study by Nishino and coworkers (7), it would be reasonable to hypothesize that furosemide inhalation would also relieve dyspnea during exercise in COPD. In the present study, we performed a double-blind, randomized, crossover study to investigate the effect of inhaled furosemide on the sensation of dyspnea during exercise in patients with COPD. As it is unknown whether inhaled furosemide has any bronchodilating effects in chronic airflow limitation, a secondary aim of this study was to investigate the effects of inhaled furosemide on spirometry of patients with COPD.
METHODS
Subjects were patients with stable COPD who satisfied the following criteria: (1) moderate to severe COPD (FEV^sub 1^
The study was conducted on 2 days (not more than 2 weeks apart) in a double-blind, randomized, crossover design. Eligible subjects were assigned to the order of study visits (placebo-furosemide or furosemide-placebo) using simple randomization. On each of the 2 days, baseline spirometry was performed before the patient was asked to inhale furosemide (4 ml of furosemide as a 10 mg/ml solution) or placebo (4 ml of 0.9% saline solution), administered by means of a jet nebulizer and nebulized to dryness with the subjects breathing tidally over 15 minutes.
Incremental cardiopulmonary exercise testing was performed immediately after completion of nebulization. After recovery from exercise and at 1 hour after nebulization of placebo or furosemide, the patients performed another spirometry. After this, the patients rested and 1 hour after termination of the incremental exercise test, they received another nebulization of furosemide or placebo (the same agent as that received earlier in the day) followed immediately by constant work rate (CWR) exercise testing at 70% of the peak work rate achieved in the initial incremental cardiopulmonary exercise testing. Figure 1 shows the timing of events and measurements during experimental visits.
At the beginning of every minute during exercise testing, each patient was asked to rate the intensity of sensation of dyspnea using a visual analog scale (VAS) (9). The analog scale consisted of a vertical straight line, 100 mm in length with 10 equally spaced markers. It was labeled 100 at the top and 0 at the bottom. Patients were instructed to point to a spot on the line indicating the sensation of respiratory discomfort at that point in time. The numerical value of zero indicated no sensation at all and 100 indicated a sensation that was intolerable. Dyspnea was defined as an unpleasant urge to breathe with no further clarification or definition given.
The primary outcome measures were dyspneic VAS scores during incremental and CWR exercise testing after inhalation of placebo or furosemide. We compared the exercise responses at standardized exercise time (SET) during incremental and during CWR exercise testing for each patient. We analyzed the data obtained during exercise testing after placebo inhalation and after furosemide inhalation using two-tailed paired t tests. SET was equal to the time of the highest equivalent amount of work (isoworkrate) or time (isotime) completed in the incremental or endurance exercise tests for each patient, that is, the time of the shorter of the two incremental or endurance tests for each patient.
Spirometry results at baseline, after placebo inhalation and after furosemide inhalation, were also analyzed using two-tailed paired t tests.
Additional details on the method for making these measurements are provided in an online supplement.
RESULTS
Of the 20 patients recruited, 8 were randomized to receiving placebo first and 12 to receiving furosemide first. Nineteen patients completed all the experimental protocols performed on 2 separate days. One patient developed acute wheezing and breathlessness after performing spirometry subsequent to inhalation of placebo on the first study visit and refused to participate further in the study. The data for this patient is excluded from analysis. The baseline anthropometric and pulmonary function data of the remaining 19 patients are shown in Table 1. All the patients had moderate or severe COPD as defined by the European Respiratory Society guidelines (10). Table 2 shows the spirometry results of patients before and after inhalation of placebo or furosemide and exercise testing. After inhalation of furosemide and exercise, there was significant improvement in mean FEV^sub 1^ and FVC compared with placebo (p = 0.038 and 0.005, respectively). There was also a significant difference in the mean FEV^sub 1^ after furosemide and exercise when compared with the mean FEV^sub 1^ after placebo and exercise (p = 0.040).
During incremental exercise testing after inhalation of placebo, 12 patients (63.2%) were ventilatory limited as they achieved all three of the following criteria: (1) a ratio of the V(dot above)E at peak exercise (V(dot above)Emax) to the maximal voluntary ventilation (MVV; calculated as FEV^sub 1^ × 40) more than 0.8, (2) MVV V(dot above)Emax less than 12 L/minute, (3) maximum heart rate more than 90% predicted. Eleven (57.9%) patients had ventilatory limitation of exercise after inhalation of furosemide. All patients who had ventilatory limitation of exercise reported dyspnea as the limiting symptom at peak exercise. The remaining patients complained of leg fatigue as the predominant symptom limiting exercise. Four patients had significant oxygen desaturation by pulse oximetry of greater than 4% during incremental exercise after placebo compared with six patients who had significant oxygen desaturation during exercise after furosemide. As shown in Table 3, none of the selected variables during peak exercise were significantly different after inhalation of placebo or furosemide. In particular, the dyspneic VAS score at peak exercise was lower after furosemide than after placebo but the difference was not statistically significant (p = 0.391). There was also no significant difference in exercise responses at SET during incremental exercise testing (see Table 4). The mean dyspneic VAS scores at SET during incremental exercise testing after inhalation of furosemide and after placebo were 35.5 ± 23.7 and 35.8 ± 22.1, respectively (p = 0.921).
At termination of CWR exercise testing, 16 patients reported dyspnea as the limiting symptom after inhalation of placebo, whereas the rest reported leg fatigue only as the predominant symptom limiting exercise. During CWR exercise testing after inhalation of furosemide, 11 patients reported dyspnea as the limiting symptom, whereas leg fatigue was the predominant symptom for the remainder. At SET during CWR exercise testing, the mean dyspneic VAS score was lower after inhalation of furosemide compared with placebo (33.7 ± 25.2 vs. 42.4 ± 24.0, respectively; p = 0.014) (Table 5). Except for three patients, all other patients had lower or similar VAS scores after inhalation of furosemide compared with placebo (see Figure 2). Although the mean duration of CWR exercise achieved by patients was longer after furosemide than after placebo (619 ± 94 vs. 572 ± 90 s, respectively), the difference was not statistically significant (p = 0.497).
Mean VAS scores over time during CWR exercise testing is shown in Figure 3. The VAS scores at the beginning, early, and intermediate stages of exercise testing were similar but differed toward the end of constant-load exercise testing.
The subjects and investigators in this study were successfully and totally blinded to the study drugs during the course of the study. None of the subjects noticed any differences during or after inhalation of furosemide and placebo. In particular, none of the subjects expressed a bitter taste in the throat, a known association after inhalation of furosemide, although this was not specifically asked of each subject. Only one patient reported a desire to urinate after incremental exercise testing after inhalation of furosemide. No other systemic or adverse effects of furosemide were noted during the study.
DISCUSSION
The main findings of this study are that inhalation of furosemide alleviates the sensation of dyspnea induced by constant-load exercise testing in patients with COPD and that there is significant bronchodilation after inhalation of furosemide compared with placebo in these patients. The finding of reduction in dyspneic sensation after inhalation of furosemide is consistent with the findings of Nishino and coworkers (7) who demonstrated that inhaled furosemide greatly alleviates the sensation of dyspnea induced experimentally by breathholding and by a combination of resistive loading and hypercapnia in normal subjects. Inhaled furosemide has also been found to suppress the behavioral response to airway occlusion in anesthetized cats without affecting their response to somatic noxious stimuli (11). These experimental findings are also in agreement with several clinical observations of nebulized furosemide being an effective and useful palliative treatment for dyspnea in patients with terminal illness. Shimoyama and Shimoyama (12) described three patients with terminal cancer having severe dyspnea in whom 20 mg of furosemide nebulized and inhaled four times a day dramatically improved the dyspnea and controlled the symptom for weeks. Another recent study (13) reported that nebulized furosemide improved dyspnea scores in 12 of 15 patients with terminal cancer whose dyspnea was uncontrollable by standard therapy. Stone and colleagues (14) also reported that inhaled furosemide caused a remarkable improvement of severe dyspnea in a patient with end-stage pulmonary Kaposi's sarcoma. Contrary to their earlier findings, Stone and Kurowska (15) recently described the initial findings of a double-blind placebo-controlled study in patients with cancer that showed no significant difference between the furosemide and saline arms for dyspnea. However, this study only investigated the effectiveness of a single dose of furosemide, rather than the regular dosing as described by Shimoyama and Kurowska in their case reports. Presently the effectiveness of nebulized furosemide for dyspnea in patients with terminal cancer has not been established.
Both incremental and constant-load exercise testing have been used to assess therapeutic responses in patients with COPD. The finding of improved dyspnea sensation after furosemide inhalation during constant-load endurance testing but not at peak exercise during incremental exercise testing and requires further elucidation. In a study aimed at testing the reproducibility of Borg dyspnea ratings, inspiratory capacity (to monitor lung hyperinflation) and endurance time during constant-load symptom-limited cycle exercise in patients with advanced COPD, O'Donnell and coworkers (16) found that Borg ratings of dyspnea measured with this exercise protocol were highly responsive to treatment with ipratropium bromide and highly reproducible when repeated over an 8-week period. In contrast, the Borg ratings at the symptom-limited peak of exercise after incremental workload testing, although highly reproducible, were poorly responsive to the inhaled anticholinergic agent. Oga and coworkers (17) also compared three different exercise tests (progressive cycle ergometry, cycle endurance test, and 6-minute walking test) and found that the endurance test was the most sensitive in detecting the effects of inhaled oxitropium bromide. Hence, dyspnea ratings during incremental exercise testing may not be as sensitive in evaluating symptom responses to therapy as ratings of dyspnea during endurance exercise testing. Another possible explanation for the positive outcome during endurance but not incremental exercise testing in this study is that the furosemide inhalation was repeated before constant-load exercise tests. Repeating the dose of inhaled furosemide before constant-load exercise tests in this study was necessary, as its effect might have diminished over the duration of the trial in each patient otherwise. Nonetheless, this could have led to a greater effectiveness of the drug in reducing dyspnea during the constant-load exercise tests. The duration of action of inhaled furosemide is not known. In the study by Nishino and coworkers (7), dyspneic sensation was measured for only 15 minutes after inhalation of 40 mg of furosemide. None of the previous studies evaluating inhaled furosemide in acute asthma (described subsequently) had monitored symptoms or lung function longer than 60 minutes. With regard to its protective effect on exercise-induced asthma, Novembre and coworkers (18) found that doubling the dose of inhaled furosemide (30 vs. 15 mg) has a longer duration of action but no difference in protective effect on exercise-induced asthma in children.
Although furosemide prevents or attenuates bronchospasm caused by many factors, such as hyperpnea, drugs (metabisulfite, bradykinin, AMP), physical agents (hypotonic and hypertonic aerosols), and allergen challenge in patients with asthma (4-6), its acute bronchodilator effect is questionable (19) and its effectiveness in acute exacerbation of asthma is unproved (20). It has also been shown that furosemide has no direct effect on the airway smooth muscle in vitro (21, 22). Several randomized, double-blind trials investigating the short-term responses of inhaled furosemide in addition to standard treatment in acute asthma exacerbation have recently been published. Pendino and coworkers (23) evaluated patients with acute asthma in the emergency department given nebulized salbutamol and either 40 mg nebulized furosemide or saline solution. No significant difference in PEF rates was detected before, 15 minutes, and 30 minutes after therapy. However, subgroup analysis of patients whose exacerbations were of short duration (
The improvement in FEV^sub 1^ after furosemide inhalation may suggest that the overall reduction in dyspneic sensation during endurance testing among the patients in this study might be accounted for by bronchodilation induced by inhaled furosemide. After inhalation of furosemide and exercise, the increase in FVC was proportionally greater than the increase in FEV^sub 1^, leading to a significant decrease in FEV^sub 1^/FVC. Assuming that the total lung capacity remained constant in each patient, the increase in FVC reflects reciprocal decrease in residual volume, i.e., the degree of hyperinflation at rest is reduced. Whether the degree of hyperinflation during exercise is also reduced is not known. As dynamic lung hyperinflation is an important causative factor of exertional dyspnea in COPD (27), future studies should include measurement of the level of lung hyperinflation at rest and during exercise. However, the V(dot above)E at SET during incremental and CWR exercise testing was not significantly different after furosemide and after placebo in this study. This observation makes the sole explanation for the alleviation of dyspnea by bronchodilation less likely although it docs not exclude the possibility that it may be a contributory factor.
Besides bronchodilation, other possible mechanisms for the alleviation of dyspnea during exercise after furosemide inhalation should also be considered. It has been postulated that inhaled furosemide has a primary effect on the airway epithelium and may influence the responsiveness of sensory nerve endings or affect the activation of inflammatory cells by inhibiting mediator release from these cells (4, 5, 28). Nishino and coworkers (7) hypothesized that the observed alleviation of dyspnea after inhalation of furosemide in healthy subjects was caused by the altered activity of sensory receptors. In particular, inhaled furosemide is considered to cause a decrease in the activity of vagal irritant and C-fiber receptors, stimulation of which increases the intensity of dyspnea and alters its quality while increasing the activity of pulmonary stretch receptors, the activation of which relieves the sensation of respiratory distress. The subsequent finding that inhaled furosemide activates pulmonary stretch receptors and inhibits the activity of pulmonary irritant receptors in anesthetized rats (29) supported this hypothesis. In this study, the observation that the effects of furosemide on reducing dyspneic sensation were mainly evident at higher levels of ventilation during constant-load exercise may be an indication of the effects of furosemide on lung receptors. The firing of the pulmonary stretch receptors increases with the degree as well as the rate of lung inflation. In fact, at higher lung volumes, the receptors are known to show more marked sensitivity to changes in flow rate than to changes in lung volume. The irritant receptor fires predominantly during inflation and its firing also depends strongly on the rate of airflow (30). In addition, activation of slowly adapting pulmonary stretch receptors is known to decrease airway cholinergic tone (31), and their stimulation by furosemide may also account for the bronchodilation observed in this study. Other postulations for the reduction of dyspneic sensation by furosemide include effects on ventilatory drive and carbon dioxide chemosensitivity. However, the VT/inspiratory time ratio and other breathing pattern parameters during exercise after furosemide or placebo were not significantly different in this study to suggest an effect on the ventilatory drive by furosemide. Minowa and coworkers (32) recently demonstrated that, in healthy subjects, inhaled furosemide improved dyspnea produced by hypercapnic hyperpnea but the mechanism of the improvement was not associated with the decrease in carbon dioxide chemosensitivity.
Although there was a reduction in dyspneic sensation at SET during endurance exercise testing after furosemide inhalation, no significant improvement in exercise endurance time was observed. In patients with COPD, ventilatory limitation is considered to be the most important reason for stopping exercise (33). As with previous studies that failed to show significant effects of bronchodilators on exercise capacity (33), a limitation of the present study was that it was unclear whether the patients were actually limited by their ventilatory capacity. Some patients could have stopped exercising due to nonventilatory reasons, such as leg muscle fatigue. In these instances, the extent to which furosemide inhalation alleviates dyspneic sensation during exercise may be irrelevant to the improvement of exercise endurance. Whereas dyspnea is more likely to be the primary limiting symptom in more advanced COPD, leg discomfort has been shown to be the predominant limiting symptom in mild to moderate disease (34). Hence, further studies should be performed on patients with more severe airflow limitation and the ratings for both dyspnea and leg fatigue during exercise should be evaluated to determine their contribution to limiting exercise.
In conclusion, inhalation of furosemide alleviates the sensation of dyspnea during constant-load exercise testing in patients with stable COPD. There is small but statistically significant improvement in mean FEV^sub 1^ after inhalation of furosemide compared with placebo in these patients.
References
1. The GOLD Expert Panel. Global strategy for the diagnosis, management and prevention of COPD. Available at: http://www.goldcopd.com. Cited July 15, 2003.
2. Gallagher CG. Exercise limitation and clinical exercise testing in chronic obstructive pulmonary disease. Clin Chest Med 1994;15:305-326.
3. Ventresca PG, Nichol GM, Barnes PJ, Chung KF. Inhaled furosemide inhibits cough induced by low-chloride solutions but not by capsaicin. Am Rev Respir Dis 1990;142:143-146.
4. Bianco S, Vaghi A, Robushi M, Pasargiklian M. Prevention of exercise-induced bronchoconstriction by inhaled furosemide. Lancet 1988;2: 252-255.
5. Bianco S, Pieroni MU, Refini KM, Rattoli L, Sestini P. Protective effect of inhaled furosemide on allergen-induced early and late asthmatic reactions. N Engl J Med 1989;321:1069-1073.
6. Robuschi M, Gambaro G, Spagnotto S, Vaghi A, Bianco S. Inhaled furosemide is highly effective in preventing ultrasonically nebulised water bronchoconstriction. Pulm Pharmacol 1987;1:187-191.
7. Nishino T, Ide T, Sudo T, Sato J. Inhaled furosemide greatly alleviates the sensation of experimentally induced dyspnea. Am J Respir Crit Core Med 2000;161:1963-1967.
8. Bestall JC, Paul EA, Garrod R, Garnham R, Jones PW, Wedzicha JA. Usefulness of the Medical Research Council (MRC) dyspnoea scale as a measure of disability in patients with chronic obstructive pulmonary diasease. Thorax 1999;54:581-586.
9. Adams L, Chronos N, Lane R, Guz A. The measurement of breathlessness induced in normal subjects: validity in two scaling techniques. Clin Sci 1985;69:7-16.
10. Siafakas NM, Vermeire P, Pride NB, Paoletti P, Gibson J, Howard P, Yernault JC, Decramer M, Higenbottam T, Postma DS, et al. Optimal assessment and management of chronic obstructive pulmonary disease (COPD). Eur Respir J 1995;8:1398-1420.
11. Nehashi S, Nishino T, Ide T. Inhaled furosemide inhibits behavioural response to airway occlusion in anesthetized cats. Anesthesiology 2001; 95:1234-1237.
12. Shimoyama N, Shimoyama M. Nebulized furosemide as a novel treatment for dyspnea in terminal cancer patients. J Pain Symptom Manage 2002; 23:73-76.
13. Kohara H, Ueoka H, Aoe K, Maeda T, Takeyama H, Saito R, Shima Y, Uchitomi Y. Effect of nebulized furosemide in terminally ill cancer patients with dyspnea. J Pain Symptom Manage 2003;26:962-967.
14. Stone P, Kurowska A, Tookman A. Nebulized furosemide for dyspnoea. Palliat Med 1994;8:258.
15. Stone P, Kurowska A. Re: nebulized furosemide for dyspnea in terminal cancer patients. J Pain Symptom Manage 2002;24:274-275.
16. O'Donnell DE, Lam M, Webb KA. Measurement of symptoms, lung hyperinflation, and endurance during exercise in chronic obstructive pulmonary disease. Am J Respir Crit Care Med 1998;158:1557-1565.
17. Oga T, Nishimura K, Tsukino M, Hajiro T, Ikeda A, Izumi T. The effects of oxitropium bromide on exercise performance in patients with stable chronic obstructive pulmonary disease. Am J Respir Crit Care Med 2000; 161:1897-1901.
18. Novembre E, Frongia G, Lomabrdi E, Resti M, Zammarchi E, Vierucci A. The preventive effect and duration of action of two doses of inhaled furosemide on exercise-induced asthma in children. J Allergy Clin Immunol 1995;96:906-909.
19. Cavaliere F, Masieri S. Furosemide protective effect against airway obstruction. Curr Drug Target 2002;3:197-201.
20. Gonzalez-Sanchez R, Trujillo-Hernandez B, Huerta M, Vasquez C, Trujillo X. Furosemide plus albuterol compared with albuterol alone in children with acute asthma. Allergy Asthma Proc 2002;23:181-184.
21. Knox AJ, Ajao P. Effect of frusemide on airway smooth muscle contractility in vitro. Thorax 1990;45:856-859.
22. Corboz MR, Ballard ST, Gao H, Benoit JN, Inglis SK, Taylor AE. Differential effects of furosemide on porcine bronchial arterial and airway smooth muscle. J Appl Physol 2000;89:1360-1364.
23. Pendino JC, Nannini LJ, Chapman KR, Slutsky A, Mollino NA. Effect of inhaled furosemide in acute asthma. J Asthma 1998;35:89-93.
24. Hinckley JB. Inhaled furosemide in the treatment of acute exacerbations of asthma. Acad Emerg Med 2000;7:1167.
25. Ono Y, Kondo T, Tanigaki T, Ohta Y. Furosemide given by inhalation ameliorates acute exacerbation of asthma. J Asthma 1997;34:283-289.
26. Rodriguez Vaxquez JC, Pino Alfonso PP, Gassiot Nuno C, Paez Prats I, Fernandez Manzano E. Assessment of the bronchodilator effect of inhaled furosemide compared to salbutamol in asthmatic patients. J Investig Alelrgol Clin Immunol 1998;8:115-118.
27. O'Donnell DE, Webb KA. Exertional breathlessness in patients with chronic airflow limitation. Am Rev Respir Dis 1993;148:1351-1357.
28. Chung KF, Barnes PJ. Loop diuretics and asthma. Pulm Pharmacol 1994; 5:1-7.
29. Sudo T, Hayashi F, Nishino T. Responses of tracheobronchial receptors to inhaled furosemide in anesthetized rats. Am J Respir Crit Care Med 2000;162:971-975.
30. Cherniack NS, Pack AI. Control of ventilation. In: Fishman AP, Elias JA, Fishman JA, Grippi MA, Kaiser LR, Senior R, editors. Fishman's manual of pulmonary diseases and disorders, 3rd ed. New York: McGraw-Hill; 1996. p. 163-176.
31. Canning BJ, Fischer A. Neural regulation of airway smooth muscle tone. Respir Physiol 2001;125:113-127.
32. Minowa Y, Ide T, Nishino T. Effects of inhaled furosemide on CO2 ventilatory responsiveness in humans. Pulm Pharmacol Ther 2002;15: 363-368.
33. Liesker JJ, Wijkstra PJ, Ten Hacken NH, Koeter GH, Potsma DS, Kerstjcns HA. A systematic review of the effects of bronchodilators on exercise capacity in patients with COPD. Chest 2002;121:597-608.
34. Killian KJ, LeBlanc P, Martin DH, Summers E, Jones NL, Campbell EJM. Exercise capacity and ventilatory, circulatory, and symptom limitation in patients with chronic airflow obstruction. Am Rev Respir Dis 1992;146:935-940.
Kian-Chung Ong, Ai-Ching Kor, Wai-Fung Chong, Arul Earnest, and Yee-Tang Wang
Department of Respiratory Medicine and Clinical Research Unit, Tan Tock Seng Hospital, Singapore
(Received in original form August 22, 2003; accepted in final form February 17, 2004)
Supported by a grant from the Tan Tock Seng Hospital Research Fund.
Correspondence and requests for reprints should be addressed to Kian-Chung Ong, M.B.B.S., F.A.M.S., F.R.C.P., Department of Respiratory Medicine, Tan Tock Seng Hospital, 11 Jalan Tan Tock Seng, Singapore 308433. E-mail: kian_chung_ong@ttsh.com.sg
This article has an online supplement, which is accessible from this issue's table of contents online at www.atsjournals.org
Am J Respir Crit Care Med Vol 169. pp 1028-1033, 2004
Originally Published in Press as DOI: 10.1164/rccm.200308 1171OC on February 20, 2004
Internet address: www.atsjournals.org
Conflict of Interest Statement: K-C.O. does not have a financial relationship with a commercial entity that has an interest in the subject of this manuscript; A-C.K. does not have a financial relationship with a commercial entity that has an interest in the subject of this manuscript; W-F.C. does not have a financial relationship with a commercial entity that has an interest in the subject of this manuscript; A.E. does not have a financial relationship with a commercial entity that has an interest in the subject of this manuscript; Y-T.W. does not have a financial relationship with a commercial entity that has an interest in the subject of this manuscript.
Copyright American Thoracic Society May 1, 2004
Provided by ProQuest Information and Learning Company. All rights Reserved