The word khat (pronounced cot) may not evoke much response from most of American society, but it could herald a significant problem for law enforcement in the near future. (1) Khat is a plant that originates in eastern Africa and the southern Middle East; people of these regions know it well and, reportedly, for centuries have chewed its leaves for their narcotic properties. Users in these areas may spend up to half of their income on the drug. (2) Khat is known by other names throughout the world; people in eastern Africa most commonly call it miraa, but it also is referred to as chat, jat, oat, kat, African salad, and Abyssinian tea. Many countries consider it a legitimate (and profitable) export.
[ILLUSTRATION OMITTED]
The United States considers khat dangerous and classifies it as a controlled substance. Undoubtedly, a passion for this drug in America's fast-paced society would present a crisis. Armed with an understanding of this natural narcotic, including its origins, chemical and medical concerns, and cultural status, law enforcement will be better equipped to combat it if it expands into the larger U.S. population.
Composition and Cultivation
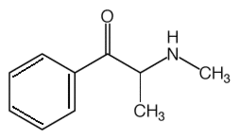
The main psychoactive ingredients in khat are cathinone (chemically similar to amphetamine) and cathine. In addition, khat plants contain chemicals called alkaloids, which long have served as narcotics and hallucinogens.
DEA classifies cathinone as a Schedule I narcotic. (3) The amounts of cathinone that exist in khat and, thus, the drug's mind-altering effects may vary based on the area where it was harvested. For instance, the amount of cathinone in khat plants from Kenya may reach 14 percent, while levels in plants from Yemen may be as low as 3.3 percent. (4) Once khat leaves dry and the cathinone evaporates, only cathine remains, and the plant drops to a Schedule IV narcotic. (5)
Current interest in cathinone exists because recent discoveries confirm that illegal laboratories produce a chemical called methcathinone, a synthetic form of cathinone. Ephedrine, a compound found in over-the-counter cold medicines, and pseudoephedrine represent the main precursor chemicals. Methcathinone, which sells as a methamphetamine alternative, commonly is called cat and often mistaken for khat. (6)
The khat tree grows 3,000 to 6,000 feet above sea level and can reach a height of 20 feet. The leaves of the plant are reddish-brown while on the tree, but quickly become a leathery yellow-green once picked. Although people may use all of the stems and leaves, they appear to prefer the young shoots at the top of the plant because they find them softer and easier to chew than the older ones toward the bottom. While most leaves are harvested for chewing, some are deliberately dried and crushed into a powder form for additional uses, such as smoking, brewing tea, or sprinkling over food. People harvest the leaves and stems of the plant on a continual basis throughout the year.
Khat leaves must be transported quickly to market because once they dry (within 48 hours), the cathinone evaporates, leaving only the milder cathine. When this happens, of course, users who crave the more potent effects find the khat of little worth. To help avoid this, harvesters package the leaves and stems in plastic bags or wrap them in banana leaves to preserve their moisture and may sprinkle them with water during transport to keep them moist. Frozen or refrigerated khat may retain cathinone longer.
Effects and Concerns
Cathinone stimulates an excessive production of dopamine, which controls feelings of pleasure and happiness, in the human brain. This can lead to a variety of short-term effects, including hallucinations, bizarre thoughts, schizophrenia, high blood pressure, rapid breathing, lethargy, mild depression, nightmares, and heightened alertness. (7) Other possible examples are a suppressed appetite for food and sex (8) and an increased ability to stay awake for long periods of time. (9) The drug also can make users more aggressive, inflate their egos, and cause them to be irrational and irritated, which may lead to unusual behavior, such as increased arguments, reckless actions, and violence. (10) Additional, less dramatic examples can include dry mouth, flushing, and an urge to urinate. (11) The effects of chewing khat, which can vary by user, may not show up for 3 or more hours, but then may last for several hours.
The long-term effects for chronic users may include anxiety, confusion, dysphoria, aggressive or agitated behavior, insomnia, high blood pressure, loss of weight due to a lack of appetite, increased heart rate and stomach irritation, and dehydration. Visual hallucinations and paranoia represent additional examples. (12) The chemicals in khat also may block the body's absorption of iron and other necessary minerals, causing potential health consequences. (13)
Advocates of khat argue that the drug eases symptoms of diabetes, asthma, and intestinal disorders. Furthermore, users claim that they are more adept at problem solving and social communication and have increased spirits and sharpened thinking. (14)
Many khat users have adapted strategies to impede the ill effects of the drug. Most veteran consumers can estimate the amount they can chew to avoid the negative effects of sleep deprivation and loss of appetite. Neophytes typically do not start chewing large amounts of khat; therefore, they build up a tolerance for the drug slowly. Some users even have supplemented alcohol or other depressants to counteract the effect that khat has on sleep. Due to the small quantity often consumed, overdose is unlikely.
While khat does have a strong psychological addiction for most users, the withdrawal symptoms are relatively minor when compared with other illegal drugs, such as heroin and cocaine. Withdrawal signs may include laziness, depression, nightmares, and slight tremors. (15) The duration of these symptoms may differ by user. Furthermore, the severity of the depression can vary and may lead to agitation or sleeping difficulties.
The rise of khat use in the United States seems to coincide with the increase in the number of immigrants arriving from eastern Africa and the Arabian Peninsula. (16) In 2000, the U.S Customs Service seized 70,008 pounds of khat, an increase of 21,070 pounds from the previous year. In Columbus, Ohio, which has the second largest Somali population in the United States, police seized 860 pounds in 2002, an increase over the previous 2 years' seizures of 633 pounds and 8.5 pounds, respectively. (17) New York City, Detroit, Minneapolis, Seattle, and San Diego may see an increase in khat arrests due to growing eastern African communities. (18)
[GRAPHIC OMITTED]
Many immigrants are unaware that khat is illegal in the United States. As a result, they often use the drug in public and later face arrest. Some cities even have seen khat advertised and sold openly in grocery stores and restaurants. Many sellers, in an attempt to keep sales of the drug quiet, only deal with users of eastern African descent and turn away everyone else.
On the street, khat currently sells for $28 to $50 a bundle (100 to 200g) and $300 to $440 a kilogram; (19) these prices currently compare with those of some other drugs, such as ecstasy and oxycodone, but are considerably lower than prices of other narcotics, such as marijuana, cocaine, and heroin. Khat's low cost makes it appealing to many drug users, especially youths.
Most of the khat enters the United States from eastern Africa via overnight shipping, although there have been some instances of cultivation in the United States. A majority of khat arrives through commercial shipping services, although some passengers aboard airlines will smuggle it in their luggage or place it with other cargo and mark it as "vegetables."
Recommendations
With khat seizures nearly doubling annually, (20) law enforcement personnel must become more cognizant of this drug. The following recommendations may assist officers to better combat its manufacturing, distribution, and use:
* Law enforcement officers must receive training in academies and in-service seminars to recognize khat. Examples of the actual drug can aid in identification. Drug detection canines also should be trained to detect khat.
* To identify users on the street, officers must have an understanding of khat symptoms and methods of use.
* Because the trafficking and smuggling practices of khat are not unlike those of other illegal narcotics coming into the United States, current drug detection methods can prove successful.
* Law enforcement agencies must use international communication and cooperation to track the progression of khat and possible dealers migrating to the United States.
* Law enforcement must be aware of any khat cultivation in the United States. The plant grows in higher altitudes with wet climates (or in greenhouses or indoors where the climate can be duplicated); in this regard, officers must monitor, as appropriate, any areas in the United States that may provide khat with the means to grow.
* Because khat has previously existed only in underdeveloped countries, few scientific studies exist. Reliable data could help law enforcement agencies understand the potential long-term effects of khat.
* To target incidents of khat use, law enforcement and medical institutions, including hospitals, research facilities, and health departments, must make a coordinated effort.
Conclusion
With khat numbers growing in the United States, law enforcement must fight this drug proactively. To evaluate the potential for khat use, Americans only need to consider the progression of other drugs, such as oxycodone and ecstasy, which started with a small community of users and now continue to spread across the country. While khat has not yet become widespread, it soon may be a legitimate problem.
If law enforcement personnel can become educated about khat, perhaps this knowledge will prove valuable when combating this drug in the United States. The key to early success in this effort is recognition.
Endnotes
(1) The authors based this article on their research and knowledge of narcotics.
(2) Teri Randall, "Khat Abuse Fuels Somali Conflict, Drains Economy," Journal of the American Medical Association 269 (January 6, 1993): 12-14.
(3) A Schedule I narcotic presents a high potential for abuse and dependence. Examples include LSD, heroin, and cocaine. For additional information, see http://www.usdoj.gov/dea/agency/csa.htm.
(4) Mwingirwa Kithure, "The Dark Side of Chewing Miraa (Khat)"; retrieved on January 23, 2004, from http://www.africaonline.com/site/Articles/1,10,2654.jsp.
(5) A Schedule IV narcotic presents a lower potential for abuse and dependence than some other controlled substances. Examples include benzodiazepines, darvon, and phenobarbital. For additional information, see http://www.usdoj.gov/dea/agency/csa.htm.
(6) U.S. Department of Justice, Drug Enforcement Administration, "Khat," Drug Intelligence Brief, June 2002; retrieved on January 23, 2004, from http://www.usdoj.gov/dea/pubs/intel/02032/02032.html.
(7) Supra note 4.
(8) Jonathan Stevenson, "Krazy Khat: Somalia's Deadly Drug War," The New Republic 207 (November 23, 1992): 17-19.
(9) Supra note 4.
(10) Supra notes 2 and 8.
(11) Richard B. Seymour, "Khat Has U.S. Presence," Psychopharmacology Update 10 (June 1999): 5.
(12) Ibid.; and Mohamed Al-Kamel, "Khat Plant"; retrieved on January 24, 2004, from http://www.geocities.com/forceps1974/khat.html.
(13) Supra note 12 (Al-Kamel).
(14) Supra note 12 (Al-Kamel).
(15) Supra note 4.
(16) "Khat Use Increases in Some U.S. Cities," Alcoholism and Drug Abuse Weekly 14 (September 16, 2002): 7-9.
(17) Ibid.
(18) Frank Bures, "From Civil War to the Drug War: Immigrants Are Risking Prison for a Taste of Home," Mother Jones 26 (November/December 2001): 23-24.
(19) Supra notes 6 and 12 (Al-Kamel).
(20) T.Trent Gegax, "Meet the Khat Heads," Newsweek, September 30, 2002, 35.
By M. Justin Crenshaw, M.S., and Tod Burke, Ph.D.
Lt. Crenshaw serves in the U.S. Air Force. Dr. Burke, a former police officer, is a professor of criminal justice at Radford University in Radford, Virginia.
COPYRIGHT 2004 Federal Bureau of Investigation
COPYRIGHT 2004 Gale Group