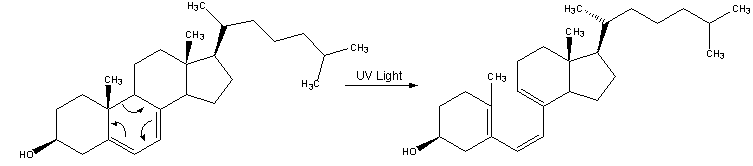
UVB-induced Conversion of 7-Dehydrocholesterol to lot,25-Dihydroxyvitamin D3 (Calcitriol) in the Human Keratinocyte Line HaCaT"
ABSTRACT
We have previously shown that keratinocytes in vitro can convert biologically inactive vitamin D3 to the hormone calcitriol. The present study was initiated to test whether ultraviolet B (UVB)-induced photolysis of provitamin D3 (7-dehydrocholesterol, [7-DHCI) which results in the formation of vitamin D3 also leads to the generation of calcitriol in keratinocytes. Submerged monolayers of HaCaT keratinocytes were preincubated with 7-DHC (25 pA,f) at 37*C and irradiated with monochromatic UVB at different wavelengths (effective UV-doses: 7.5-60 mj/ cm2), or a narrow-band fluorescent lamp Philips TL-01 (UVB-doses: 125-1500 mJ/cm2). Irradiation with both sources of UVB resulted in the generation of different amounts of previtamin Dj in our in vitro model followed by time-dependent isomerization to vitamin D3 and consecutive formation of calcitriol in the picomolar range. Unirradiated cultures or cultures exposed to wavelengths >315 mm generated no or only trace amounts of calcitriol. The conversion of vitamin D3 generated after UVB irradiation to calcitriol is inhibited by ketoconazole indicating the involvement of P450 mixed function oxidases in this chemical reaction. The generation of calcitriol was wavelength- and UVB dose dependent and reached approximately 18-fold higher levels after irradiation at 297 nm than at 310 nm (effective UVB dose: 30 mJ/cmz). Hence, keratinocytes may be a potential source of biologically active calcitriol within epidermis, when irradiated with therapeutical doses of UVB.
INTRODUCTION
Calcitriol (la,25-dihydroxyvitamin D3, [lu,25(OH)2D3Dt, the most potent biologically active form of vitamin D3 (VD3) is produced by a cascade of reactions including photochemical VD3 synthesis in the skin and subsequent hydroxylation at the C-25 atom in the liver and at C-lot position in the kidney (1). Calcitriol and other vitamin D analogs have antiproliferative and prodifferentiative effects on epidermal keratinocytes (2-4) and have become potent therapeutical agents for the treatment of proliferative skin disorders such as psoriasis. It has been shown that cultured keratinocytes can convert exogenous calcidiol (25-hydroxyvitamin D3 [250HD3]) to calcitriol (5-7) and we have previously shown that both keratinocytes from the HaCaT cell line (8) and from human skin equivalents can convert exogenous VD3 to calcitriol (9,10). This implicates that functionally active lotand 25-hydroxylases are present in keratinocytes. Provitamin D3 (7-dehydrocholesterol [7-DHC]) exposed to ultraviolet B (UVB) radiation, converts in vivo (11) and in vitro (12) to previtaminD3 (pre-VD3), which in turn isomerizes to VD3. Until now, the complete pathway from 7-DHC to la,25(OH)2D3 has neither been found in the skin nor in cultures of epidermal skin cells. It was the aim of this study to provide evidence that photolysis of 7-DHC could result in the formation of la,25(OH)2D3 in cultured HaCaT keratinocytes.
MATERIALS AND METHODS
Chemicals and reagents. Dulbecco modified Eagle medium (DMEM) and fetal calf serum (FCS) were provided from GIBCO (Eggenstein, Germany). Culture dishes ( 30 mm) were from Falcon (Heidelberg, Germany). la-OHD3 and la,25(OH)2D3 were kindly provided by Hoffmann-La Roche AG (Basel, Switzerland). 25-hydroxy[26,27-methyl-3H]vitamin D3 (3H-25OHD3, 177 Ci/mmol), la,25-dihydroxy[26,27-methyl-3H]vitamin DI (3H-Ils,25(OH)2D3, 173,5 Ci/mmol) and 24R,25-dihydroxy[26,27-methyl-3H]vitamin D3 (3H-24R,25(OH)2D3, 170 Ci/mmol) were purchased from Amersham (Braunschweig, Germany). The la,25(OH)2D3-radioreceptor assay kit from Nichols Institute (Bad Nauheim, Germany) was used. VD3, solvents for high performance liquid chromatography (HPLC) (nhexane, 2-propanol and methanol) were provided by Merck (Darmstadt, Germany). Lumisterol and tachysterol were donated by Dr. A. Kissmeyer (Leo Pharmaceutical Products, Ballerup, Denmark). Ketoconazole was bought from Paesel & Lorei (Frankfurt/M, Germany). Bovine serum albumin (BSA), purity -99% (product number A 0281) and 7-DHC were from Sigma (Deisenhofen, Germany). Scintillation cocktail, Ready Protein+, was purchased from Beckman Instruments (Fullerton, CA). NO-bis trimethylsilyl acetamide, Ntrimethylsilylimidazole and trimethylchlorosilane were purchased from Macherey-Nagel (Dueren, Germany). Pre-VD3 was prepared by thermal treatment of an ethanolic solution of VD3 (1.0 itg/mL) at 60*C for 16 h under nitrogen according to Isler and Brubacher (13). VD3 (retention time: 10.57 min) and generated pre-VD3 (retention time: 8.22 min) were separated by normal phase (NP)-HPLC (eluent 1, see "HPLC analysis"). Pre-VD3 was fractionated from 8.00 to 9.00 min. The UV spectrum of pre-133 (X.,,: 260 nm and km;": 230 nm) was identical to that described in the literature (14).
Cell culture protocol. HaCaT cells (used with the permission of Prof. N. E. Fusenig of the German Cancer Research Center, Heidelberg) were seeded at a density of 5 x 104 cells/cmz in culture dishes (o 30 mm) and grown in DMEM supplemented with 5% (vol/ vol) FCS. Cultures were maintained at 37*C and 5% COz in air. After 2 days the medium was replaced for 18 h by serum-free DMEM in order to induce synchronization of the cell cycle. The serum-free DMEM was then replaced by FCS-supplemented medium for 8 h, followed by serum-free DMEM supplemented with 1.0% (wt/vol) of highly purified BSA (Sigma). At this time cells were preconfluent (equal to -0.7 X 106 cells/dish). Cell numbers and their viability were assessed using a CASY' 1-cell counter (Scharfe System GmbH, Reutlingen, Germany). The viability was always ?93%.
Incubation conditions. 7-DHC (30 nmol) dissolved in 6 liL ethanol, was added to the medium (1.2 mL) of the cell cultures. Control experiments were carried out (1) in the presence of 7-DHC without irradiation; (2) in the absence of 7-DHC with and without irradiation; and (3) in cell-free culture medium containing 7-DHC with irradiation. After UVB irradiation and incubation in dark the medium and detached cells were separately extracted with methanol:chloroform (1:1). The chloroform phases were used for the determination of pre-VD3, VD3 and 101,25(OH)ZDj.
UV irradiation. Samples were exposed to UVB emitted by (1) a tuneable high intensity monochromator (part of the DERMOLUM UM Fa. Miller Optik-Elektronik, Moosinning, Germany); or (2) a fluorescence tube Philips TL-01 (Fa. Philips, Eindhooven; The Netherlands): the so-called "narrow-band UVB lamp." The TL-01 lamp is characterized by a spectral output of 74% in the narrow waveband 311-313 nm. The spectral irradiance ET(X) of the TL-01 tube is depicted in Fig. 1. Using (1) the high intensity monochromator (full wavelength at half maximum: 5 nm) an irradiance Ee = 0.28 mW/ cm2 (inhomogeneity within the irradiation spot of o 15 turn; - 10%) was measured at the bottom of the culture dish. The irradiance was controlled by a thermopile TS 50-1 (Physikalisch-Technische Werkstatten, Jena, Germany), calibration with PTB (Braunschweig, Germany). The culture dish (i 30 mm) was continuously rotated round the axis. The UV-spot (i 15 mm) was positioned at the radius of the rotating dish. The UVB doses used were adapted to these experimental conditions (effective dose = Def = applied UVB dose X 0.25). The irradiance of (2) the TL-01 lamp at the bottom of the culture dish was measured as Ee = 0.86 mW/cmz by an adapted photometer UV-Meter (Fa. Waldmann, Villingen-Schwenningen, Germany).
HPLC analysis. NP-HPLC: Merck/Hitachi; column: LiChroCART 250-4, Superspher Si 60, 5 Rm; solvent system 1 (n-hexane: 2-propanol = 95:5 [vol/vol]; flow rate: 0.5 mL/min) for the determination of pre-VD3, VD3 and 7-DHC; solvent system 2 (n-hexane: 2-propanol:methanol = 87:10:3 [vol/vol/vol]; flow rate: 1.0 mL/min) for separation of calcitriol. The peaks of pre-VD3 and VD; (retention times: 8.22 and 10.57 min, respectively) in the former system were quantified by UV detection at 265 nm. The peak homogeneity was checked using a diode array detector (L-4500, Merck/Hitachi). Fractions containing calcitriol were collected and analyzed for calcitriol by a radioreceptor assay. The results were converted to picomoles of 1,25(OH)2D3 and normalized to 106 cells. Reversed phase (RP)HPLC: the calcitriol generated was identified by cochromatography of the 3H-labeled standard using a Hibar column, 250-4, LiChrospher IOORP-18, 5 wm, (Merck, Darmstadt, Germany); solvent system 3: methanol:water = 85:15 (vol/vol), flow rate: 1 mL/min. Calcitriol-containing fractions were analyzed for their content of calcitriol.
Gas chromatography-mass spectrometry. The dried fractions from HPLC were derivatized to the trimethylsilyl ether derivatives and analyzed by gas chromatography-mass spectrometry (GC-MS). The derivatized sample (1 wL) was directly and manually injected into a model 5890/II gas chromatograph equipped with a 25 m X 0.2 mm HP-1 capillary column (crosslinked methylsiloxane, 0.33 Itm) and interfaced with a model 5989A MS-Engine (Hewlett-Packard, Palo Alto, CA). GC conditions were the following: carrier gas, helium; column head pressure, 10 psi; injector temperature, 260*C; oven temperature gradient, maintained at 200*C for 1 min, increased to 260*C at 30C/min, then increased to 300'C at 20'C/min, held at 300*C for 10 min; interface temperature 300'C. Electron impact (EI) MS conditions were the following: source temperature, 250'C; analyzer temperature, 120*C; energy, 70 eV. All samples were run in triplicate under the control of the HP-ChemStation data system. For selective ion monitoring, the most abundant ions at m/z 452 and m/ z 501 were monitored for lI,25(OH)2D3.
Statistical analysis. Results are presented as mean or mean + standard deviation. Data were analyzed by one-way analysis of variance (Bonferroni method).
RESULTS
UVB-induced conversion of 7-DHC to VD3
In order to monitor UVB-induced photolysis under experimental conditions, 7-DHC in 1.2 mL culture medium was exposed to monochromatic UVB light in absence of cells. Figure 2 shows several chromatograms (mobile phase 1) obtained from extracts of DMEM containing 25 tLM 7-DHC and supplemented with 1.0% BSA at increasing incubation times after UVB irradiation at 297 2.5 nm and Deft = 30 mJ/cmz. The photolyzed product pre-VD3 was identified by cochromatography of pre-VD3 prepared by thermal isomerization of VD3 as described in "Materials and Methods" and by its UV spectrum (Xmax: 260 nm and Vim;": 230 nm). VD3 formed was identified by cochromatography of the VD3 reference substance and by its UV spectrum (Amax: 265 nm and Xmin: 229 nm). The photoisomers of pre-VD3, lumisterol and tachysterol (retention times: 9.00 and 10.96 min, respectively), were not detectable after irradiation. The time courses of the UVB-induced conversion of 7-DHC via preVD3 to VD3 in DMEM without cells and with cells after irradiation at 297 + 2.5 run and Deff = 30 mJ/cmz indicate that the isomerization of pre-VD3 to VD3 is completed after 16-20 h (data not shown in detail). We failed to detect endogenous 7-DHC in HaCaT cells. Figure 3 indicates that the VD3 synthesis is dependent on the wavelength of UV radiation in the presence and absence of cells. There is a shift of the wavelength corresponding to maximum synthesis rates of VD3 from 294 nm in DMEM without cells to 303 nm in presence of cells. The maximum amounts of VD3 generated in medium alone and in regular cultures were almost comparable. After irradiation at 320 nm no VD3 production was detectable in both the presence and absence of cells.
To investigate the effect of cellular transport of 7-DHC on the overall results cell cultures were incubated with 25 IiM 7-DHC; after 1 h the medium was removed, cells were washed with 1.2 mL phosphate buffered saline (PBS) twice and reconstituted with 1.2 mL PBS. The cell cultures were then irradiated at 297 nm (D,ff = 30 mJ/cmz), incubated for 16 h at 37'C in dark and extracted as described. The cellular uptake of 7-DHC was measured to be approximately 20% of the total amount of 7-DHC (30 nmol) prior added to the cell culture. The corresponding cellular VD3 level was about 37% of the total amount of VD3 (977 348 pmol, N = 3) formed in the original cell culture.
Conversion of photosynthesized VD3 to lat,25(OH)2D3 in HaCaT cells
Fractions (1-25) obtained after NP-HPLC (solvent system 2) of extracts of irradiated cell cultures (297 2.5 run, Drf = 30 mJ/cm2) and of nonirradiated controls were analyzed for calcitriol (Fig. 4). The peak in fractions 20-22 is identical with calcitriol. Minor peaks at 7 and 17 min have not been identified yet. The radioactivity peaks of 3H-25OHD3 and 3H24R(OH)2D3 appear after 7 and 12 min, respectively. However, we failed to detect 250HD3 using NP-HPLC and UV detection at 265 nm (data not shown). No calcitriol was detectable in analogous fractions of nonirradiated cultures (Fig. 4) as well as in the other controls (ii and iii, not shown). Calcitriol comigrated with synthetic 3H-la,25(OH)2D3 in both NP- and RPHPLC systems. To exclude the presence of 19-nor-, 10-keto25-hydroxyvitamin D3 (19-nor, 10-keto-25OHD3), which comigrates with la,25(OH)2D3 in NP-HPLC, 300 pg putative calcitriol was separated by RP-HPLC and the amount of calcitriol recovered was about the same as injected initially. This indicates the absence of 19-nor, 10-keto-25OHD3 which should be separable from calcitriol under these chromatographic conditions.
Studies of the TMS derivatives of synthetic and generated calcitriol by GC-MS demonstrated identical retention times. Two resulting peaks (pyro- and isopyro derivative) were identically seen for synthetic and presumptive la,25(OH)2D3. The full-scan El mass spectra of the pertrimethylsilyl ether derivatives from the reference compound and presumptive lot,25(OH)2D3 (pyro peak) are depicted in Fig. SA,B. The mass spectra produced by the pyro- and isopyro peaks differ only in the relative abundance of the mass fragments. Of note, weak molecular ions [M+] at in/z 632 can be observed in both spectra (no mass peaks of intensity greater than 9% of the base peak). The spectra showed the most abundant ion at ml z 452 QM-180]) resulting from the loss of two silanol groups from the ionized molecule. There were two other prominent ions at m/z 542 ([+-90]) resulting from the loss of one silanol group and at mlz 362 ([M*-270]) arising from the loss of three silanol groups. The intense ion at mIz 501 ([M+- 13 131]) may arise from the loss of the C2,3,4-3-silanol fragment. The peak at m/z 131 corresponded to a secondary propyl ether trimethylsilyl ion obtained by the cleavage of the bond between C24 and C25.
The addition of ketoconazole (1, 5 and 10 pAP to cultures of HaCaT cells immediately after irradiation at 297 - 2.5 nm and Def = 30 mJ/cm2, and further incubation for 16 h in the dark caused a dose-dependent inhibition (Fig. 6) of the formation of la,25(OH)2D3. The antioxidant, 1,2-dianilinoethane (10 pAf), only had a marginal inhibitory effect (315 run (data not shown). The UVB dose-response relationship depicted in Fig. 7B indicates a good linear correlation of the VD3 levels and the respective amounts of calcitriol (y = 2.15 X -33.9, R = 0.97) up to Deft = 30 mJ/cmz in this cell system, after a constant incubation time of 16 h. While VD3 levels increase in a linear manner up to Deff = 60 mJ/cm2, calcitriol synthesis is maximal at Derr = 30 mJ/cm2 and slightly declines at higher UV-doses. The decrease in viability of cells after irradiation was time and dose dependent (data not shown in detail). For instance, cell viability decreased from 91 2.1%, N = 3 (unirradiated control) to 44.5 3.9%, N = 3, after irradiation at 297 nm (Derr = 60 mJ/cm2) followed by a 16 h incubation time.
To estimate the conversion rate of intracellularly generated VD3 to calcitriol, cells were washed with PBS as previously described. Cells treated in this manner produced approximately 37% of the total amount of calcitriol (1024 199 fmol/106 cells; N = 3) generated in the regular cell culture.
Experiments using the TL-01 lamp as UVB source showed a time course of VD3 and calcitriol formation in cultures of HaCaT cells similar to that with monochromatic UVB at 297 nm (Fig. 7C vs A). The synthesis of VD3 shows no further increase after 6 h, and calcitriol formation in keratinocytes is maximal at near 16 h after irradiation. The photoisomers of pre-VD3, lumisterol and tachysterol (retention times: 9.00 and 10.96 min, respectively), were undetectable under these irradiation conditions. In contrast to the VD3 concentration, the calcitriol level significantly declines from 16 to 24 h. Notably, the maximum concentration of VD3 was only 35% of that obtained with monochromatic light at 297 nm. The maximum amount of calcitriol generated was only 16% of that obtained in comparable experiments using UVB at 297 run. The UVB dose-response relationship depicted in Fig. 7D indicates a maximum of the calcitriol synthesis at 250 mJ/cm2 after a constant incubation time of 16 h. In contrast, VD3 levels linearly increase up to doses of about 630 mJ/cm2 and show a plateau at higher UVB doses. The viability of cells time- and dose-dependently decreased after irradiation (data not shown in detail). For instance, cell viability decreased from 91 2.1%, N = 3 (unirradiated control) to 38.0 + 2.1%, N = 3, after irradiation at a dose of 1500 mJ/cm2 followed by 16 h incubation time.
Figure 8 demonstrates, that HaCaT cells preincubated with 25 pM 7-DHC and irradiated with (1) monochromatic light at 297 2.5 nm and 310 2.5 nm (D,ff = 30 mJ/ cm2); and (2) UVB from the narrow-band TL-01 lamp (dominated by a strong and narrow peak around 312 nm) generated very different amounts of calcitriol. Notably, the monochromatic light at 310 nm initiated the synthesis of only small amounts of calcitriol (--50.1 pmol/106 cells) as compared to the TL-01 lamp (-0.7 pmol/106 cells).
DISCUSSION
UVB light causes photolysis of 7-DHC to pre-VD3, which undergoes a rearrangement of its double bond structure to form the thermodynamically more stable VD3 (11,12). Previously we have shown that cultured keratinocytes can hydroxylate exogenous VD3 to la,25(OH)2D3 (9,10). In this study we investigated the generation of la,25(OH)2D3 from VD3 produced after UVB-induced photolysis of the precursor 7-DHC by HaCaT keratinocytes.
We first established the UVB-induced nonenzymatic isomerization of 7-DHC via pre-VD3 to VD3 in the absence and presence of HaCaT cells. The identities of generated pre-13 and VD3 were confirmed by NP-HPLC as well as by their UV spectra. Our results obtained under cell-free conditions are in accordance with earlier findings which show that UVB light (optimum wavelengths between 295 and 300 nm) causes photolysis of 7-DHC to pre-13 in human skin (11,15) and in cultures of human keratinocytes (12). The isomerization of pre-VD3 to VD3 is a time- and temperaturedependent process (11,15,16). Our results indicate that the conversion of pre-VD3 to VD3 at 370C in the presence of HaCaT cells is almost completed after 16 h. This is comparable to human skin where this isomerization needs approximately 20 h (16). Notably, there is a shift of the wavelength corresponding to maximum synthesis rates of VD3 from 294 nm in DMEM without cells to 303 nm in the presence of HaCaT cells. This observation is in contrast to earlier findings (11,12,15) which demonstrated that wavelengths between 295 and 300 nm optimally photolyze 7DHC to pre-133 in human skin (11,15) and in cultures of human keratinocytes (12). We have no explanation for this discrepancy at present. On the other hand, our results are very similar to observations made in rat skin (17), where the optimum wavelength for the synthesis of VD3 (305 nm) was longer than in organic solvents (295 run). It is a fact that both kinetics and thermodynamics of the reaction pre-VD3 VD3 may change significantly in many anisotropic microenvironments (16,18). Based on a liposomal model that mimics the cutaneous generation of VD3 it has been hypothesized by Tian and Holick (19) that cellular membrane phosholipids which interact with pre-VD3 stabilize pre-VD3 in its cZc-conformation, the only conformer that can convert to VD3. The stronger these interactions are, the more pre-13 is in its cZc-conformation and the faster is the rate of its conversion to VD3. It is conceivable that these phospholipidpre VD3 interactions are influenced by wavelength-dependent photodegradation of membrane phospholipids resulting in different rates of VD3 formation. Such wavelength-dependent processes also might explain the changeability of VD3 production between 285 and 295 nm. In agreement with literature (15), we found no synthesis of VD3 at wavelengths >315 nm.
These results show for the first time that VD3 generated by UVB-induced photolysis of 7-DHC is further metabolized to la,25(OH)2D3 in cultured HaCaT cells. Interestingly, two other metabolites were found which possessed crossreactivity with 1(x,25(OH)2D3 in the radioreceptor assay. The identity of the two metabolites is not known at the present time. The isolated putative calcitriol was clearly identified as 1t,25(OH)2D3 by both NP- and RP-HPLC as well as by GCMS. The mass spectrum was identical with that of the reference substance and followed a fragmentation pattern as previously described (20-22). In control experiments without cells using medium alone or in unirradiated controls with cells no calcitriol was detected in the culture extracts. Thus, the generation of calcitriol requires the presence of HaCaT cells as well as a UVB-induced VD3 synthesis.
Studies using ketoconazole, a cytochrome P450 enzyme inhibitor (23), clearly implicated the involvement of cytochrome P450 mixed-function oxidases in the formation of la,25(OH)2D3. 1,2-Dianilinoethane (10 VJP, a known radical scavenger and antioxidant (24) in contrast, exerted only a slight quench effect on the hydroxylations of VD3. These and our previous results argue in favor of the presence of both la-and 25-hydroxylase activity in HaCaT cells.
The time course of the UVB-induced generation of calcitriol in HaCaT cells shows a maximum at 16 h incubation time, followed by a gradual decrease. Calcitriol probably induces its own catabolism to calcitrioic acid (25) during this time period. Inhibition of lot-hydroxylase by lcL,25(OH)2D3 (5,7), may also contribute to a reduction in generation of calcitriol.
We found that monochromatic UVB dose-dependently (Dew = 7.50.0 mJ/cm2) induced generation of 10x,25(OH)2D3 from VD3, which was formed after photoconversion of 7DHC at 297 2.5 run in our in vitro system. For comparison, an effective dose of 30 mJ/cm2 at 297 -+ 2.5 nm corresponds to -1.5 minimal erythemal dose (MED) for hypopigmented Caucasian skin. The concentration of 7-DHC used in our experiments (30 nmol/1.2 mL medium equivalent to 4.25 nmol/cm2 cell monolayer) was comparable to the level of 7-DHC (=2.7 nmol/cm2) found in hypopigmented Caucasian human leg skin (11) and in human neonatal foreskin (-6.1 nmol/cmz) (26). The production rate of calcitriol showed a positive correlation to the amount of VD3 generated after UVB irradiation at 297 run up to De = 30 mJ/cm2. Apparently, UVB inactivates intracellular hydroxylases at Deft - 30 mJ/cm2, while nonenzymatic synthesis of VD3 remains unaffected. It should be noted that the conversion rate of VD3 obtained by photolysis of 7-DHC to calcitriol was approximately two to five-fold higher than that of comparable concentrations of added VD3 (9). We assume that the UV induced extra- and intracellular generation of VD3 contributes to a very efficient metabolism to calcitriol more than in cells which depend on the diffusion of extracellular VD3 through the cell membrane.
The TL-01 lamp is commonly used in the phototherapeutical treatment of psoriasis (27-30). A radiant exposure of about 1000 mJ/cmz emitted from the TL-01 lamp corresponds to 1.0 MED for hypopigmented Caucasian human skin. UVB radiation from the TL-01 produced less VD3 and calcitriol in our in vitro model than monochromatic light at 297 nm. Interestingly, monochromatic light at 310 * 2.5 nm induced the formation of considerably less amounts of VD3 and calcitriol compared to the narrow-band TL-01 lamp. The differences in the spectral output of the lamps could explain these differences. As depicted in Fig. 1, the TL-01 lamp emits, in addition to the strong and narrow peak around 312 mn, a number of smaller peaks at 305 and 365 run as well as minor peaks at 282, 292, 297, 327 and 334 run. This is in accordance with data of the relative spectral energy distribution of the TL-01 lamp published by Van Weelden et aL (27). We speculate that the small peak at 305 nm and/or the minor peaks at 292 and 297 run are responsible for the stronger effects on the VD3 pathway compared to monochromatic UVB at 310 nm. UVB from the TL-01 lamp apparently inactivates intracellular hydroxylases at doses above 250 mJ/cmz, while the nonenzymatic synthesis of VD3 remains unaffected up to 630 mJ/cm2. The plateau of VD3 observed at doses -630 mJ/cm2 is not caused by UVB-induced isomerization of pre-13 to lumisterol or tachysterol which are increasingly generated after extended UVB irradiation (31). We failed to detect these isomers at doses between 630 and 1500 mJ/cm2 in our cell system. However, it cannot be excluded that at doses -630 mJ/cmz a part of VD3 generated within the irradiation time is photolyzed to 5,6trans-VD3, suprasterol 1 or suprastrrol 2 (32). Furthermore, it is not clear whether UVB radiation has direct regulatory effects on the activity of lot- and/or 25-hydroxylase in HaCaT cells. More recently, we have found that vitamin D3 25-hydroxylase messenger RNA is not constitutively expressed in normal human keratinocytes, but induced by both monochromatic UVB at 300 nm and VD3 (33). This finding implicates regulatory effects of UVB radiation on the activity of intracellular VD3 hydroxylase(s).
In conclusion, our findings demonstrate that the generation of calcitriol is predominantly determined by the VD3 level produced after photolysis of 7-DHC. One remaining question is whether UVB-induced synthesis of calcitriol can explain the effectiveness of UVB phototherapy in the treatment of psoriasis.
Acknowledgements-The skillful technical assistance of Mrs. A. Kampf is gratefully acknowledged. The authors thank Drs. W. Dorr and Tai C. Chen for critical reading of the manuscript.
Posted on the website on 1 November 2000.
tAbbreviations: BSA, bovine serum albumin; 7-DHC, 7-dehydrocholesterol; DMEM, Dulbecco modified Eagle medium; Deft, effective UVB dose; Ee, irradiance; El, electron impact; FCS, fetal
calf serum; GC-MS, gas chromatography-mass spectrometry; MED, minimal erythemal dose; NP-HPLC, normal phase-high performance liquid chromatography; la-011133, lo-hydroxyvitamin 133; la,25(OH)2D3, la,25-dihydroxyvitamin 133; 250HD3, 25hydroxyvitamin 133; 19-nor, 10-keto-250HD3, 19-nor-, 10-keto25-hydroxyvitamin 13; PBS, phosphate buffered saline; pre-VD3, previtamin D3; RP, reversed phase; UVB, ultraviolet B; VD3, vitamin D3.
REFERENCES
1. Haussler, M. R. (1986) Vitamin D receptors: nature and function. Annu. Rev. Nutr. 6, 527-562.
2. Hosomi, J., J. Hosoi, E. Abe, T. Suda and T. Kuroki (1983) Regulation of terminal differentiation of cultured mouse epidermal cells by lot,25-dihydroxyvitamin D3. Endocrinology 113, 1950-1957.
3. Smith, E. L., N. C. Walworth and M. F. Holick (1986) Effect of la.,25-dihydroxyvitamin D3 on the morphologic and biochemical differentiation of cultured human epidermal keratinocytes grown in serum-free conditions. J. Investig. Dermatol. 86, 709-714.
4. McLane, J. A., M. Katz and N. Abdelkader (1990) Effect of 1,25-dihydroxyvitamin D3 on human keratinocytes grown under different culture conditions. In Vitro Cell. Dev. Biol. 26, 379387.
5. Bikle, D. D., M. K. Nemanic, E. Gee and P. Elias (1986) 1,25Dihydroxyvitamin D3 production by human keratinocytes. J. Clin. Investig. 78, 557-566.
6. Matsumoto, K., Y. Azuma, M. Kiyoki, H. Okumura, K. Hashimoto and K. Yoshikawa (1991) Involvement of endogenously produced 1,25-dihydroxyvitamin D-3 in the growth and differentiation of human keratinocytes. Biochim. Biophys. Acta 1092, 311-318.
7. Lehmann, B. (1997) HaCaT cell line as a model system for vitamin Dj metabolism in human skin. J. Investig. DernatoL 108, 78-82.
8. Boukamp, P., R. T. Petrussevska, D. Breitcreutz, J. Hornung, A. Marham and N. E. Fusenig (1988) Normal keratinization in a spontaneously immortalized aneuploid human keratinocyte cell line. J. Cell. Biol. 106, 761-771.
9. Lehmann, B., J. Pietzsch, A. Kampf and M. Meurer (1998) Human keratinocyte line HaCaT metabolizes la hydroxyvitamin D3 and vitamin D3 to lIa,25-dihydroxyvitamin D3 (calcitriol). J. Dermatol. Sci. 18, 118-127.
10. Lehmann, B., T. Rudolph, J. Pietzsch and M. Meurer (2000) Conversion of vitamin D3 to lu,25-dihydroxyvitamin D3 in human skin equivalents. Exp. Dermatol. 9, 97-103.
11. Holick, M. F., J. A. MacLaughlin, M. B. Clark, S. A. Holick and J. T. Potts (1980) Photosynthesis of previtamin D3 in human skin and the physiologic consequences. Science 210, 203-205.
12. Nemanic, M. K., J. Whitney and P. M. Elias (1985) In vitro synthesis of vitamin D-3 by cultured human keratinocytes and fibroblasts: action spectrum and effect of AY-9944. Biochim. Biophys. Acta 841, 267-277.
13. Isler, 0. and G. Brubacher (1982) Vitamine 1. Fettlisliche Vitamine. 2. Die D-Vitamine. Georg Thieme, Stuttgart.
14. Holick, M. F., J. E. Frommer, S. C. McNeal, N. M. Richtand, J. W. Henley and J. T. Potts (1977) Photometabolism of 7dehydrocholesterol to previtamin D. in skin. Biochem. Biophys. Res. Commun. 76, 107-115.
15. MacLaughlin, J. A., R. R. Anderson and M. F. Holick (1982) Spectral character of sunlight modulates photosynthesis of previtamin D3 and its photoisomers in human skin. Science 216, 1001-1003.
16. Tian, X. Q., T. C. Chen, L. Y. Matsuoka, J. Wortsman and M. F. Holick (1993) Kinetik and thermodynamic studies of the conversion of previtamin D3 to vitamin D3 in human skin. J. BioL Chem. 268, 14 888-14 892.
17. Takada, K. (1983) Formation of fatty acid esterified vitamin D3 in rat skin by exposure to ultraviolet radiation. J. Lipid Res. 24, 441-448.
18. Tian, X. Q. and M. F. Holick (1995) Catalyzed thermal isomerization between previtamin D3 and vitamin D3 via [-cyclodextrin complexation. J. Biol. Chem. 270, 8706-8711.
19. Tian, X. Q. and M. F. Holick (1999) A liposomal model that mimics the cutaneous production of vitamin D3. J. Biol. Chem. 274, 4174-4179.
20. Poon, P. M. K., Y. T. Mak and C. P. Pang (1993) Gas chromatographic-mass fragmentographic determination of serum lot,25 dihydroxyvitamin D3. Clin. Biochem. 26, 461-469.
21. Schroeder, N. J., D. J. H. Trafford, J. Cunningham, G. Jones and H. L. J. Makin (1994) In vivo dibydrotachysterol2 metabolism in normal man: la- and Ip-hydroxylation of 25-hydroxydihydrotachysterol2 and effects on plasma parathyroid hormone and la,25-dihydroxyvitamin D3 concentrations. J. Clin. Endocrinol. Metab. 78, 1481-1487.
22. Mawer, E. B., M. E. Hayes, S. E. Heys, M. Davies, A. White, M. F. Stewart and G. N. Smith (1994) Constitutive synthesis of 1,25-dihydroxyvitamin D3 by a human small cell lung cancer cell line. J. Clin. Endocrinol. Metab. 79, 554-560.
23. Wilkinson, C. F., K. Hetnarski, G. P. Cantwell and F. J. DiCarlo (1974) Structure-activity relationships in the effects of 1 alkylimidazoles on microsomal oxidation in vitro and in vivo. Biochem. Pharmacol. 23, 2377-2386.
24. Sietsema, W. K. and H. F. DeLuca (1982) Retinoic acid 5,6epoxidase: properties and biological significance. J. Biol. Chem. 257, 4265.
25. Ray, S., R. Ray and M. F. Holick (1995) Metabolism of 3Hlot,25-dihydroxyvitamin D3 in cultured human keratinocytes. J. Cell. Biochem. 59, 117-122.
26. Obi-Tabot, E. T., X. Q. Tian, T. C. Chen and M. F. Holick (2000) A human skin equivalent model that mimics the photoproduction of vitamin D3 in human skin. In Vitro Cell. Dev. Biol. 36A, 201-204.
27. Van Weelden, H., H. Baart de la Faille, E. Young and J. C. van der Leun (1988) A new development in UVB phototherapy of psoriasis. Br. J. Dermatol. 119, 11-19.
28. Green, C., J. Ferguson, T. Lakshmipathi and B. E. Johnson (1988) 311 nm UVB phototherapy-an effective treatment for psoriasis. Br. J. Dermatol. 119, 691-696.
29. Karvonen, J., E. L. Kokkonen and E. Ruotsalainen (1989) 311 nm UVB lamps in the treatment of psoriasis with the Ingram Regimen. Acta Dermatol. Venereol. (Stockh.) 69, 82-85.
30. Larko, 0. (1989) Treatment of psoriasis with a new UVB-lamp. Acta Dermatol. Venereol. (Stockh.) 69, 357-359.
31. Holick, M. F., J. A. MacLaughlin and S. H. Doppelt (1981) Regulation of cutaneous previtamin D3 photosynthesis in man: skin pigment is not an essential regulator. Science 211, 590593.
32. Webb, A. R., B. de Costa and M. F. Holick (1989) Sunlight regulates the cutaneous production of vitamin D3 by causing its photodegradation. J. Clin. EndocrinoL Metab. 68, 882-887.
33. Lehmann, B., 0. Tiebel and M. Meurer (1999) Expression of vitamin D3 25-hydroxylase (CYP27) mRNA after induction by vitamin D3 or UVB radiation in keratinocytes of human skin equivalents-a preliminary study. Arch. Dermatol. Res. 291, 507-510.
Bodo Lehmann*, Peter Knuschke and Michael Meurer
Department of Dermatology, Carl Gustav Carus Medical School, Dresden University of Technology, Germany Received 8 September 2000; accepted 19 September 2000
*To whom correspondence should be addressed at: Carl Gustav Carus Medical School, Dresden University of Technology, Department of Dermatology, Fetscherstrasse 74, D-01307 Dresden, Germany. Fax: 49-351-458-4338;
e-mail: bodo.lehmann@mailbox.tu-dresden.de
Copyright American Society of Photobiology Dec 2000
Provided by ProQuest Information and Learning Company. All rights Reserved