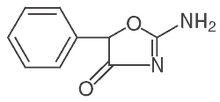
Cylert
Pemoline (sold as Cylert®) is a medication used to treat attention-deficit hyperactivity disorder (ADHD). Under the Convention on Psychotropic Substances, it is a Schedule IV drug . more...
Pemoline has some advantages over other stimulants in that it doesn't reduce the appetite or cause dry mouth. However, in some patients Pemoline is suspected of causing liver damage, so regular liver tests are performed are those treated with it. Since receiving FDA aproval in 1975, it has been linked with 21 cases of liver failure, of which 13 resulted in liver replacement or death. In 2005, the FDA withdrew approval for Pemoline due to pressure from certain public advocacy groups. Patient support group the Narcolepsy Network tried to persuade the FDA not to ban it, on the grounds that some patients find all other treatments ineffective or to have dehabilitating side effects. The medication was used by an estimated 10,000 Americans afflicted with narcolepsy.
Read more at Wikipedia.org