CASE REPORT
Possible Dangerous Interaction of Oxycontin and Carisoprodol
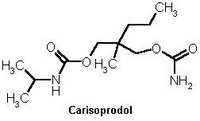
TO THE EDITOR: Oxycontin is a time-release-controlled formulation of oxycodone that can be taken every 12 hours for the effective treatment of pain caused by cancer and other types of chronic pain. However, abuse of oxycontin is common; no other prescription drug in the past 20 years has been so widely misused so soon after its release. (1) Carisoprodol (Soma) is a skeletal muscle relaxant that also has shown potential for abuse. (2) We report a case of a possible dangerous interaction of Oxycontin and carisoprodol.
A 49-year-old woman received treatment for severe degenerative disease of the lumbar spine with 40 mg of Oxycontin twice daily for over a year. Although she continued to complain of pain and paravertebral muscle spasm, her physician was reluctant to increase the dosage of Oxycontin. One tablet (350 mg) of carisoprodol four times daily was prescribed. After taking this regimen for a week without relief she increased the dosage to eight to 10 tablets a day, assuming this would be safe. Her boyfriend called an ambulance after finding her lying unconscious on the floor and was unable to awaken her.
An examination revealed blood pressure of 130/60 mm Hg and respirations of 14 per minute and decreased in depth. She was responsive only to painful stimuli. Her pupils were small but equal and reactive. She was given 2 mg of naloxone intravenously with rapid return of alertness. A count of her remaining supply of medications verified that she had not taken any extra Oxycontin and a drug screen was negative except for opiates.
The most likely explanation for this patient's adverse reaction would be a response to the additive depressant effects on the central nervous system (CNS) of both Oxycontin and carisoprodol. Although the effects of Oxycontin are well known in the medical community, many physicians are unaware that carisoprodol is metabolized to meprobamate (Miltown), which is a Class IV controlled substance with significant physiologic effects, and has potential for abuse and dependency. (3) Previous reports (4,5) have suggested that substance abusers may use carisoprodol to augment or modify the effects of illicit drugs and that combinations of carisoprodol and tramadol (Ultram) may have potent psychotropic effects.
Carisoprodol and Oxycontin should only be prescribed concomitantly with extreme caution because of the abuse potential and the potentially dangerous additive CNS-depressant effect of the two drugs. While numerous deaths related to Oxycontin have occurred, (1) it should be noted that a retrospective review (6) of autopsy cases in Jefferson County, Alabama from January 1, 1986 to October 31, 1997 found carisoprodol present in 24 cases; the reviewers concluded that the drug was probably partly responsible for those deaths. (6) The interactions of carisoprodol with other CNS depressant drugs appears to be particularly significant. Carisoprodol has been made a controlled substance in the state of Alabama (6) and may eventually be made a controlled substance in other states or at the federal level.
ROY R. REEVES, D.O., PH.D.
JAMES E. MACK, PH.D.
University of Mississippi School of Medicine
VA Medical Center
1500 E. Woodrow Wilson Dr.
Jackson, MS 39216
REFERENCES
(1.) Charatan F. Time-release analgesic drug causes fatal overdoses in United States. West J Med 2001;175:82.
(2.) Littrell RA, Hayes LR, Stillner V. Carisoprodol (Soma): a new and cautious perspective on an old agent. South Med J 1993;86:753-6.
(3.) Littrell RA, Sage T, Miller W. Meprobamate dependence secondary to carisoprodol (Soma) use. Am J Drug Alcohol Abuse 1993;19:133-4.
(4.) Reeves RR, Carter OS, Pinkofsky HB. Use of carisoprodol by substance abusers to modify the effects of illicit drugs. South Med J 1999;92:441.
(5.) Reeves RR, Liberto V. Abuse of combinations of carisoprodol and tramadol. South Med J 2001; 94:512-4.
(6.) Davis GG, Alexander CB. A review of carisoprodol deaths in Jefferson County, Alabama. South Med J 1998;91:726-30.
Send letters to Jay Siwek, M.D., Editor, American Family Physician, 11400 Tomahawk Creek Pkwy., Leawood, KS 66211-2672; fax: 913-906-6080; e-mail: afplet@ aafp.org. Please include your complete address, telephone number, and fax number. Letters should be submitted on disk, double-spaced, fewer than 500 words, and limited to one table or figure and six references. Please submit a word count. Letters submitted for publication in AFP must not be submitted to any other publication. Possible conflicts of interest must be disclosed at time of submission. Submission of a letter will be construed as granting the AAFP permission to publish the letter in any of its publications in any form. The editors may edit letters to meet style and space requirements.
COPYRIGHT 2003 American Academy of Family Physicians
COPYRIGHT 2003 Gale Group