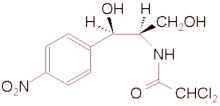
During an investigation, 55 biotype C (bovine and caprine biotype) Staphylococcus aureus isolated from 43 cows suffering from mastitis, and 20 biotype C Staph. aureus strains from the nares and the side of nail-tips of the right thumbs of 20 farm workers (milkers and animal attendants) on six small dairy farms in Assam and Meghalaya were isolated. Three strains from the former and six strains from the latter from among the isolates on two of the farms were found resistant to chloramphenicol, when tested with a routine susceptibility test. Test of the organisms by the agar dilution method indicated that the resistant strains had a minimal inhibitory concentration for chloramphenicol of 32 (mu)g/ml or more, while, the chloramphenicol sensitive strains and two reference strains, Staph. aureus ATCC 25923 and Micrococcus luteus ATCC 9341, had
Key words Chloramphenicol inactivation - chloramphenicol resistance - Staphylococcus aureus biotype C
Chloramphenicol resistant staphylococci were reported prior to 19661. Inactivation of chloramphenicol by Escherichia coli was reported first in 1967. The antibiotic is inactivated by some resistant organisms by acetylation with acetyltransferase, which is acquired through the process of development of chromosomal resistance against chloramphenicol;. Inactivation of chloramphenicol by chloramphenicol resistant staphylococci has been reported from our laboratory's. However, these are reports on staphylococci isolated from animals.
Considering that chloramphenicol is an important and widely used antibiotic, and that, Staphylococcus aureus is a versatile organism, not only capable of affecting man and animals equally, but also capable of cross-infection between man and animals, the present investigation was undertaken to ascertain whether Staph. aureus, resistant to chloramphenicol and isolated from man and lactating animals on dairy farms, could inactivate the antibiotic or not.
A total of 58 strains of Staph. aureus from 43 cows suffering from mastitis, and another 21 strains of Staph. aureus from the nares and the inner side of nail tips of the right thumbs of 21 farm workers (milkers and animal attendants) on three dairy farms in the Khanapara region of Assam and another three farms in the Ribhoi district of Meghalaya were isolated during 1996-1998 .^sup 6^
The Staph. aureus were differentiated from micrococci with the help of Baird-Parker's classification^sup 7^. The strains were biotyped at the Laboratory of Veterinary Bacteriology, State University of Ghent, Belgium by a simplified method8.
Two reference stains, Staph. aureus ATCC 25923 and Micrococcus luteus ATCC 9341 (R-1 and R-2, respectively), were procured, from the American Type Culture Collection (ATCC), Rockville, USA. Agar disc diffusion test9 was carried out in Diagnostic Sensitivity Test agar (Hi Media, Mumbai), using discs, among others, of chiorarnphenicol (30 (mu)g/disc).
The minimal inhibitory concentration (MIC) of chloramphenicol for the chloramphenicol resistant biotype C strains, two chloramphenicol sensitive strains (one each from human [H-S] and bovine [B-S] origins), and two reference strains, R- and R-2, were determined by agar dilution method10. The Gots test" was carried out in 1.8 per cent Penassay Seed agar (Difco Laboratories, USA), using M luteus ATCC 9341 as the seed strain. Tests (quantitative) for inactivation of chloramphenicol were carried out by a standard method 12.
After biotyping, 55 of the 58 bovine strains and 20 of the 21 human strains of Staph. aureus were found to be of biotype C. The remaining strains were unclassifiable. Susceptibility tests indicated that three of the 55 bovine strains and six of the 21 human strains were resistant to chloramphenicol.
The results of the MIC tests indicated that one bovine strain and one human strain had an MIC of chloramphenicol of 32 (mu)g/ml, while, two bovine strains and five human strains had MIC>64 gg/ml, confirming that all of them were resistant to chloramphenicol. Strains B-S and H-S had MIC of 8 gg/ml, R-1 had 4 gg/ml, and R-2 had an MIC of I gg/mI, all these strains were considered as susceptible to chloramphenicol. One strain each of bovine and human origin and resistant to chloramphenicol showed negative results in the Gots test and two bovine and five human strains showed positive results. The results of antimicrobial assay for inactivation of chloramphenicol are shown in the Table. After 36 h of culture, chloramphenicol could not be detected (the minimum detectable limit was 0.48 gg/ml) in the culture tubes of the strains which showed positive results in the Gots test.
In our earlier study^sup 5^, 10.07 per cent of the chloramphenicol resistant Staph. aureus strains of animal origin showed positive results in the Gots test. However, the strains were not biotyped. Resistance to chloramphenicol has been reported to be commonly associated with resistance to penicillin G, ampicillin, tetracycline HCI, erythromycin, and lincomycin6,13. However, the reverse was not usually found.
It was reported in 1968 that when chloramphenicol was administered orally to sheep, therapeutic levels of chloramphenicol were not attained in serum apparently due to its inactivation by the microbial flora of the rumen14. However, in case of erythromycin, an in vitro erythromycin resistant Lactobacillus fermentum strain, experimentally colonized in the crops of gnotobiotic chicks, inactivated orally administered therapeutic dosages of erythromycin in vivo, resulting in subtherapeutic levels of the antibiotic in blood of chicks". It is, therefore, possible that a bacterial strain, which can inactivate chloramphenicol in vitro, may also be able to inactivate chloramphenicol in vivo.
Acknowledgment
The first and the third authors are grateful to Dr M. Debackere, formerly. Dean. Faculty of Veterinary Medicine, State University of Ghent, Belgium and the Director of the Laboratories for Pharmacology. Pharmacy. and Toxicology, Belgium for providing material to carry out the study. The authors thank Dr L.A. Devriese, Laboratory of Veterinary Bacteriology, Faculty of Veterinary Medicine, State University of Ghent, Belgium for biotyping the Staph. aureus strains.
References
1. Suzuki Y. Okamoto S, Kono M. Basis of chloramphenicol resistance in naturally isolated resistant staphylococci. J Bacteriol 1966: 92 : 798-9.
2. Shaw WV. The enzymatic acetylation of chloramphenicol by extracts of R factor-resistant Escherichia coli. JBiol Chem 1967; 242: 687-93.
3. Philpott-Howard J. Antibiotic resistance and Haemophilus influenzae. J Antimicrob Chemother 1984; 13: 199-200.
4. Buragohain J. Dutta GN. Inactivation of chloramphenicol by chloramphenicol-resistant staphylococci from bovine subclinical mastitis. Indian Vet J 1990; 67 : 493-4.
5. Buragohain J, Dutta GN. Inactivation of chloramphenicol by staphylococci isolated from mastitic cows. Indian J Vet Med 1997; 17: 14-6.
6. Gogoi J. Public health importance of Staphylococcus aureus isolated from cases of bovine mastitis and milkers. M V Sc. thesis. Guwahati : Assam Agricultural University, 1998.
7. Collins CH, Lyne PM. Microbiological methods. 3rd ed. London: Butterworths, Baltimore: University Park Press; 1970 p. 276-81.
8. Devriese LA. A simplified system for biotyping Staphylococcus aureus strains isolated from different animal species. J Appl Bacterial 1984: 56: 215-20.
9. Matsen JM, Barry AL. Susceptibility testing: diffusion test procedures. In: Lenette EH, Spaulding EH, Traunt JP, editors. Manual of clinical microbiology. 2nd ed. Washington DC American Society for Microbiology; 1974 p. 418-27.
10. Washington JA, Barry AL. Dilution susceptibility tests: agar and macro-broth dilution procedures. In: Lennette EH, Balows A, Hausler WJ, Truant JP. editors, Manual of clinical microbiology. 3rd ed. Washington DC : American Society for Microbiology; 1980 p.453-8.
11. Gots JS. The detection of pen icil lipase-producing properties of ,microorganisms. Science 1945; 102: 309.
12. Sabath LD, Anhalt JP. Assay of antimicrobics. In: Lennettee EH, Balows A, Hausler WJ, Truant iP, editors. Manual 'of clinical microbiology, 3rd ed. Washington DC : American Society for Microbiology; 1980 p. 485-90.
13. Buragohain J, Dutta GN. Antibiotic resistance patterns among isolates from bovine mammary origin. Indian./Comp Microbiol Immunol Infect Dis 1998; 19: 110-3.
14. Theodorides VJ, DiCuollo CJ, Guarini JR. Pagano JF. Serum concentrations of chloramphenicol after intraruminal and intra-abomasal administration in sheep. Am J 1,"et Res 1968, 29 : 643-5.
15. Devriese LA, Dutta GN. Effects of erythromycin-inactivating Lactobacillus crop flora on blood levels of erythromycin given orally to chicks. J Vet Pharmacol Ther 1984; 7: 49-53.
G.N. Dutta, Jully GoGoi* & Jyoti Buragohain
Department of Epidemiology & Preventive Medicine, College of Veterinary Science Assam Agricultural University, Guwahati, India
Accepted November 23, 2000
Present address : *Assistant Professor, Veterinary Public Health, College of Veterinary Sciences, Central Agricultural University, Aizawl, Mizoram
Reprint requests: Dr Gunindra N. Dutta, Department of Epidemiology & Preventive Medicine, Assam Agricultural University Khanapara Campus. Guwahati 781022, India
Copyright Indian Council of Medical Research Jan 2001
Provided by ProQuest Information and Learning Company. All rights Reserved